A fully automated high-throughput workflow for 3D-based chemical screening in human midbrain organoids
- PMID: 33138918
- PMCID: PMC7609049
- DOI: 10.7554/eLife.52904
A fully automated high-throughput workflow for 3D-based chemical screening in human midbrain organoids
Abstract
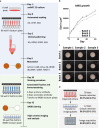
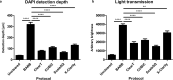
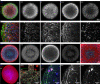
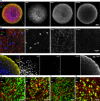
Three-dimensional (3D) culture systems have fueled hopes to bring about the next generation of more physiologically relevant high-throughput screens (HTS). However, current protocols yield either complex but highly heterogeneous aggregates ('organoids') or 3D structures with less physiological relevance ('spheroids'). Here, we present a scalable, HTS-compatible workflow for the automated generation, maintenance, and optical analysis of human midbrain organoids in standard 96-well-plates. The resulting organoids possess a highly homogeneous morphology, size, global gene expression, cellular composition, and structure. They present significant features of the human midbrain and display spontaneous aggregate-wide synchronized neural activity. By automating the entire workflow from generation to analysis, we enhance the intra- and inter-batch reproducibility as demonstrated via RNA sequencing and quantitative whole mount high-content imaging. This allows assessing drug effects at the single-cell level within a complex 3D cell environment in a fully automated HTS workflow.
Keywords: 3D cell culture; automation; forebrain; high content imaging; high throughput screening; human; midbrain; organoid; regenerative medicine; stem cells.
Plain language summary
In 1907, the American zoologist Ross Granville Harrison developed the first technique to artificially grow animal cells outside the body in a liquid medium. Cells are still grown in much the same way in modern laboratories: a single layer of cells is placed in a warm incubator with nutrient-rich broth. These cell layers are often used to test new drugs, but they cannot recapitulate the complexity of a real organ made from multiple cell types within a living, breathing human body. Growing three-dimensional miniature organs or 'organoids' that behave in a similar way to real organs is the next step towards creating better platforms for drug screening, but there are several difficulties inherent to this process. For one thing, it is hard to recreate the multitude of cell types that make up an organ. For another, the cells that do grow often fail to connect and communicate with each other in biologically realistic ways. It is also tough to grow a large number of organoids that all behave in the same way, making it hard to know whether a particular drug works or whether it is just being tested on a 'good' organoid. Renner et al. have been able to overcome these issues by using robotic technology to create thousands of identical, mid-brain organoids from human cells in the lab. The robots perform a series of precisely controlled tasks – including dispensing the initial cells into wells, feeding organoids as they grow and testing them at different stages of development. These mini-brains, which are the size of the head of a pin, mimic the part of the brain where Parkinson's disease first manifests. They can be used to test new drugs for Parkinson's, and to better understand the biology of the brain. Perhaps more importantly, other types of organoids can be created using the same technique to model diseases that affect other areas of the brain, or other organs altogether. For example, Renner et al. also generated forebrain organoids using an automated approach for both generation and analysis. This research, which shows that organoids can be grown and tested in a fully automated, reproducible and scalable way, creates a platform to quickly, cheaply and easily test thousands of drugs for Parkinson's and other difficult-to-treat diseases in a human setting. This approach has the potential to reduce research waste by increasing the chances that a drug that works in the lab will also ultimately work in a patient; and reduce animal experiments, as drugs that do not work in human tissues will not proceed to animal testing.
© 2020, Renner et al.
Conflict of interest statement
HR, MG, MO, HS, JB The work presented in this study is the subject of the patent application EP 18 19 2698.0-1120 to the European Patent Office, where HR, MG, MO, HRS, and JMB are inventors. KB, TK, JW, SP, DZ, YT, PD, JK, SL, GS No competing interests declared
Figures
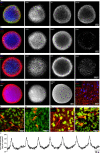

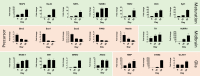
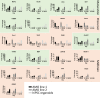
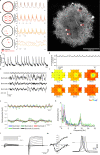
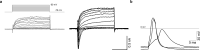


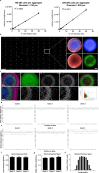
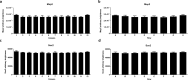

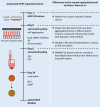
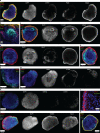
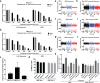
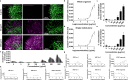
References
-
- Abud EM, Ramirez RN, Martinez ES, Healy LM, Nguyen CHH, Newman SA, Yeromin AV, Scarfone VM, Marsh SE, Fimbres C, Caraway CA, Fote GM, Madany AM, Agrawal A, Kayed R, Gylys KH, Cahalan MD, Cummings BJ, Antel JP, Mortazavi A, Carson MJ, Poon WW, Blurton-Jones M. iPSC-Derived human Microglia-like cells to study neurological diseases. Neuron. 2017;94:278–293. doi: 10.1016/j.neuron.2017.03.042. - DOI - PMC - PubMed
-
- Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, Harris MA, Hill DP, Issel-Tarver L, Kasarskis A, Lewis S, Matese JC, Richardson JE, Ringwald M, Rubin GM, Sherlock G. Gene ontology: tool for the unification of biology the gene ontology consortium. Nature Genetics. 2000;25:25–29. doi: 10.1038/75556. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

