Malformation of Tear Ducts Underlies the Epiphora and Precocious Eyelid Opening in Prickle 1 Mutant Mice: Genetic Implications for Tear Duct Genesis
- PMID: 33141892
- PMCID: PMC7645213
- DOI: 10.1167/iovs.61.13.6
Malformation of Tear Ducts Underlies the Epiphora and Precocious Eyelid Opening in Prickle 1 Mutant Mice: Genetic Implications for Tear Duct Genesis
Abstract
Purpose: Obstruction of the tear drainage causes a range of ocular surface disorders. Hitherto, the genetics of tear duct development and obstruction has been scarcely explored, and related animal models are lacking. This study aims to study the potential role of the Wnt/PCP pathway mediated by Prickle 1 in tear duct development and diseases.
Methods: A severe hypomorphic Prickle 1 mutant was generated. Histology and immunohistochemistry were performed to compare wild type, Prickle 1 hypomorphic, and null mutant tear ducts. In situ hybridization was conducted to identify the signaling components in the developing tear ducts. Three-dimensional (3D) reconstruction was used to detect the human embryonic tear duct.
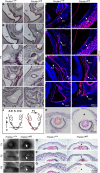
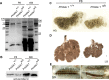
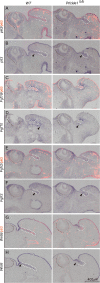
Results: Here, we report that a severe Prickle 1 hypomorph mouse line exhibited epiphora. This phenotype was due to the blockage of the tear drainage by incompletely formed nasolacrimal duct (NLD) and lacrimal canaliculi (LC), which also causes precocious eyelid opening. We observed a dose-dependent requirement of Prickle 1 for tear duct outgrowth. An investigation of the expression of Wnt/PCP core genes demonstrated a subset of PCP signaling components expressed in the developing tear duct. Furthermore, Prickle 1 is not required for the expression of Fgfr2/Fgf10 and p63 genes, which are associated with the NLD and LC hypoplasia in humans. Last, we showed that Prickle 1 expression in the developing tear drainage system is conserved between mice and humans.
Conclusions: The study suggests that malformed tear ducts caused by disruption of Prickle 1 underlies the epiphora and precocious eyelid opening.
Conflict of interest statement
Disclosure:
Figures




Similar articles
-
Obstruction of the Tear Drainage Altered Lacrimal Gland Structure and Function.Invest Ophthalmol Vis Sci. 2023 Jul 3;64(10):13. doi: 10.1167/iovs.64.10.13. Invest Ophthalmol Vis Sci. 2023. PMID: 37440262 Free PMC article.
-
A Spatiotemporal Requirement for Prickle 1-Mediated PCP Signaling in Eyelid Morphogenesis and Homeostasis.Invest Ophthalmol Vis Sci. 2018 Feb 1;59(2):952-966. doi: 10.1167/iovs.17-22947. Invest Ophthalmol Vis Sci. 2018. PMID: 29450535
-
Ocular surface pathogenesis associated with precocious eyelid opening and necrotic autologous tissue in mouse with disruption of Prickle 1 gene.Exp Eye Res. 2019 Mar;180:208-225. doi: 10.1016/j.exer.2018.12.012. Epub 2018 Dec 24. Exp Eye Res. 2019. PMID: 30590023
-
[Epiphora. Age-related changes of the ocular surface, eyelid function and the efferent tear ducts].Ophthalmologe. 2009 Mar;106(3):229-34. doi: 10.1007/s00347-008-1907-8. Ophthalmologe. 2009. PMID: 19280197 Review. German.
-
The human nasolacrimal ducts.Adv Anat Embryol Cell Biol. 2003;170:III-XI, 1-106. Adv Anat Embryol Cell Biol. 2003. PMID: 12645158 Review.
Cited by
-
Evaluating the repeatability of the multifocal electroretinogram in the cone dominant 13-lined ground squirrel.Doc Ophthalmol. 2025 Jun;150(3):117-126. doi: 10.1007/s10633-025-10010-9. Epub 2025 Mar 11. Doc Ophthalmol. 2025. PMID: 40067596 Free PMC article.
-
Obstruction of the Tear Drainage Altered Lacrimal Gland Structure and Function.Invest Ophthalmol Vis Sci. 2023 Jul 3;64(10):13. doi: 10.1167/iovs.64.10.13. Invest Ophthalmol Vis Sci. 2023. PMID: 37440262 Free PMC article.
-
Expansion of genotypic and phenotypic findings in ADAMTS18-related ocular pathology.Am J Ophthalmol Case Rep. 2025 Jan 16;37:102262. doi: 10.1016/j.ajoc.2025.102262. eCollection 2025 Mar. Am J Ophthalmol Case Rep. 2025. PMID: 39902400 Free PMC article.
-
Cerebrospinal Fluid Pressure Reduction Induces Glia-Mediated Retinal Inflammation and Leads to Retinal Ganglion Cell Injury in Rats.Mol Neurobiol. 2023 Oct;60(10):5770-5788. doi: 10.1007/s12035-023-03430-8. Epub 2023 Jun 22. Mol Neurobiol. 2023. PMID: 37347366
-
Single-Cell Characterization of the Frizzled 5 (Fz5) Mutant Mouse and Human Persistent Fetal Vasculature (PFV).Invest Ophthalmol Vis Sci. 2023 Mar 1;64(3):8. doi: 10.1167/iovs.64.3.8. Invest Ophthalmol Vis Sci. 2023. PMID: 36867129 Free PMC article.
References
-
- Frame NJ, Burkat CN.. Identifying an appropriate animal model for the nasolacrimal drainage system. Ophthalmic Plast Reconstr Surg. 2009; 25: 354–358. - PubMed
-
- Thiessen D. Harderian Gland: Porphyrin Metabolism, Behavioral and Endocrine Effects. In: Webb SM, Hoffman RA, Puig-Domingo ML, Reiter RJ, eds. The function of the Harderian gland in the Mongolian Gerbil, Meriones unguiculatus. Berlin, Germany: Springer; 1992: 127–140.
-
- Burling KMC, Da Silva Curiel J, Koblik P, Bellhorn RW. Anatomy of the rabbit nasolacrimal duct and its clinical implications. Prog Vet Compar Ophthalmol. 1991; 1: 33–40.
-
- Paulsen F, Thale A, Schaudig U. [Nasolacrimal ducts and the dry eye]. Ophthalmologe. 2002; 99: 566–574. - PubMed
-
- Paulsen FP, Foge M, Thale AB, Tillmann BN, Mentlein R. Animal model for the absorption of lipophilic substances from tear fluid by the epithelium of the nasolacrimal ducts. Invest Ophthalmol Vis Sci. 2002; 43: 3137–3143. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

