The Novel Herbal Cocktail AGA Alleviates Oral Cancer through Inducing Apoptosis, Inhibited Migration and Promotion of Cell Cycle Arrest at SubG1 Phase
- PMID: 33142749
- PMCID: PMC7692053
- DOI: 10.3390/cancers12113214
The Novel Herbal Cocktail AGA Alleviates Oral Cancer through Inducing Apoptosis, Inhibited Migration and Promotion of Cell Cycle Arrest at SubG1 Phase
Abstract
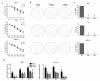
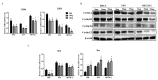
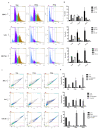
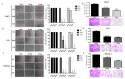
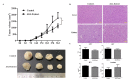
Traditional Chinese medicines Antler's extract (A) and Ganoderma lucidum (G) and Antrodia Camphorata (A) have been known to individually contain a plethora of bioactive factors including triterpenoids, polysaccharides etc., exerting various curative impacts such as anti-inflammatory, anti-oxidative, anti-atherosclerotic and anti-viral activities. However, their combinatorial therapeutic efficacy for oral cancer has not been investigated. Hence, we synthesized a robust cocktail called AGA and investigated its anti-oral cancer potential in vitro and in vivo. An MTT assay revealed the IC50 of AGA to be about 15 mg at 72 h. Therefore, 10 mg and 20 mg doses were selected to study the effect of AGA. The AGA significantly inhibited proliferation of oral cancer cells (HSC3, SAS, and OECM-1) in a dose- and time-dependent manner. AGA retarded cell cycle regulators (CDK4, CDK6, cyclin A, B1, D1 and E2) and apoptosis inhibitory protein Bcl-2, but enhanced pro-apoptotic protein Bax and a higher percentage of cells in Sub-G1 phase. Mechanistically, AGA suppressed all EMT markers; consequently, it decreased the migration ability of cancer cells. AGA significantly reduced xenograft tumor growth in nude mice with no adverse events in liver and renal toxicity. Conclusively, AGA strongly inhibited oral cancer through inducing apoptosis and inhibiting the migration and promotion of cell cycle arrest at subG1 phase, which may be mediated primarily via cocktail-contained triterpenoids and polysaccharides.
Keywords: Antler’s extract; Antrodia Camphorata; Ganoderma lucidum; anti-oxidation; cell cycle; oral cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
AGA induces sub-G1 cell cycle arrest and apoptosis in human colon cancer cells through p53-independent/p53-dependent pathway.BMC Cancer. 2023 Jan 2;23(1):1. doi: 10.1186/s12885-022-10466-x. BMC Cancer. 2023. PMID: 36597025 Free PMC article.
-
The ethanol extracts of sporoderm-broken spores of Ganoderma lucidum inhibit colorectal cancer in vitro and in vivo.Oncol Rep. 2017 Nov;38(5):2803-2813. doi: 10.3892/or.2017.6010. Epub 2017 Sep 27. Oncol Rep. 2017. PMID: 29048673 Free PMC article.
-
Chloroform Extract of Solanum lyratum Induced G0/G1 Arrest via p21/p16 and Induced Apoptosis via Reactive Oxygen Species, Caspases and Mitochondrial Pathways in Human Oral Cancer Cell Lines.Am J Chin Med. 2015;43(7):1453-69. doi: 10.1142/S0192415X15500822. Epub 2015 Oct 18. Am J Chin Med. 2015. PMID: 26477797
-
Ganoderiol F purified from Ganoderma leucocontextum retards cell cycle progression by inhibiting CDK4/CDK6.Cell Cycle. 2019 Nov;18(21):3030-3043. doi: 10.1080/15384101.2019.1667705. Epub 2019 Sep 22. Cell Cycle. 2019. PMID: 31544588 Free PMC article.
-
Ethanol extract of Antrodia camphorata inhibits proliferation of HCT-8 human colorectal cancer cells by arresting cell cycle progression and inducing apoptosis.Mol Med Rep. 2017 Oct;16(4):4941-4947. doi: 10.3892/mmr.2017.7207. Epub 2017 Aug 10. Mol Med Rep. 2017. PMID: 28849005
Cited by
-
AGA induces sub-G1 cell cycle arrest and apoptosis in human colon cancer cells through p53-independent/p53-dependent pathway.BMC Cancer. 2023 Jan 2;23(1):1. doi: 10.1186/s12885-022-10466-x. BMC Cancer. 2023. PMID: 36597025 Free PMC article.
-
Physicochemical Transformations of Silver Nanoparticles in the Oro-Gastrointestinal Tract Mildly Affect Their Toxicity to Intestinal Cells In Vitro: An AOP-Oriented Testing Approach.Toxics. 2023 Feb 21;11(3):199. doi: 10.3390/toxics11030199. Toxics. 2023. PMID: 36976964 Free PMC article.
-
Exploring the Potential of Natural Product-Based Nanomedicine for Maintaining Oral Health.Molecules. 2022 Mar 7;27(5):1725. doi: 10.3390/molecules27051725. Molecules. 2022. PMID: 35268826 Free PMC article. Review.
-
Trametes robiniophila Murr Sensitizes Gastric Cancer Cells to 5-Fluorouracil by Modulating Tumor Microenvironment.Front Pharmacol. 2022 May 17;13:911663. doi: 10.3389/fphar.2022.911663. eCollection 2022. Front Pharmacol. 2022. PMID: 35656301 Free PMC article.
-
Ethnomycological Investigation in Serbia: Astonishing Realm of Mycomedicines and Mycofood.J Fungi (Basel). 2021 Apr 29;7(5):349. doi: 10.3390/jof7050349. J Fungi (Basel). 2021. PMID: 33947042 Free PMC article.
References
-
- Hung L.C., Kung P.T., Lung C.H., Tsai M.H., Liu S.A., Chiu L.T., Huang K.H., Tsai W.C. Assessment of the Risk of Oral Cancer Incidence in A High-Risk Population and Establishment of A Predictive Model for Oral Cancer Incidence Using A Population-Based Cohort in Taiwan. Int. J Environ. Res. Public Health. 2020;17:665. doi: 10.3390/ijerph17020665. - DOI - PMC - PubMed
-
- Liu H.Y., Huang C.F., Li C.H., Tsai C.Y., Chen W.H., Wei H.J., Wang M.F., Kuo Y.H., Cheong M.L., Deng W.P. Osteoporosis Recovery by Antrodia camphorata Alcohol Extracts through Bone Regeneration in SAMP8 Mice. Evid Based Complement. Alternat. Med. 2016;2016:2617868. doi: 10.1155/2017/6297946. - DOI - PMC - PubMed
-
- Gu L., Mo E., Yang Z., Fang Z., Sun B., Wang C., Zhu X., Bao J., Sung C. Effects of Red Deer Antlers on Cutaneous Wound Healing in Full-thickness Rat Models. Asian Australas J. Anim. Sci. 2008;21:277–290. doi: 10.5713/ajas.2008.70348. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

