Dysbindin deficiency Alters Cardiac BLOC-1 Complex and Myozap Levels in Mice
- PMID: 33142804
- PMCID: PMC7692170
- DOI: 10.3390/cells9112390
Dysbindin deficiency Alters Cardiac BLOC-1 Complex and Myozap Levels in Mice
Abstract
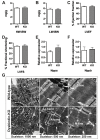
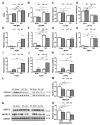
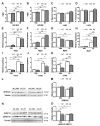
Dysbindin, a schizophrenia susceptibility marker and an essential constituent of BLOC-1 (biogenesis of lysosome-related organelles complex-1), has recently been associated with cardiomyocyte hypertrophy through the activation of Myozap-RhoA-mediated SRF signaling. We employed sandy mice (Dtnbp1_KO), which completely lack Dysbindin protein because of a spontaneous deletion of introns 5-7 of the Dtnbp1 gene, for pathophysiological characterization of the heart. Unlike in vitro, the loss-of-function of Dysbindin did not attenuate cardiac hypertrophy, either in response to transverse aortic constriction stress or upon phenylephrine treatment. Interestingly, however, the levels of hypertrophy-inducing interaction partner Myozap as well as the BLOC-1 partners of Dysbindin like Muted and Pallidin were dramatically reduced in Dtnbp1_KO mouse hearts. Taken together, our data suggest that Dysbindin's role in cardiomyocyte hypertrophy is redundant in vivo, yet essential to maintain the stability of its direct interaction partners like Myozap, Pallidin and Muted.
Keywords: Dysbindin; Muted; Myozap; Pallidin; cardiac hypertrophy.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures

References
-
- Yalcin F., Kucukler N., Cingolani O., Mbiyangadu B., Sorensen L., Pinherio A., Abraham M.R., Abraham T.P. Evolution of ventricular hypertrophy and myocardial mechanics in physiological and pathological hypertrophy. J. Appl. Physiol. 2019;126:354–362. doi: 10.1152/japplphysiol.00199.2016. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

