The Role of Molecular Chaperones in Virus Infection and Implications for Understanding and Treating COVID-19
- PMID: 33143379
- PMCID: PMC7693988
- DOI: 10.3390/jcm9113518
The Role of Molecular Chaperones in Virus Infection and Implications for Understanding and Treating COVID-19
Abstract
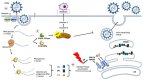
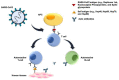
The COVID-19 pandemic made imperative the search for means to end it, which requires a knowledge of the mechanisms underpinning the multiplication and spread of its cause, the coronavirus SARS-CoV-2. Many viruses use members of the hosts' chaperoning system to infect the target cells, replicate, and spread, and here we present illustrative examples. Unfortunately, the role of chaperones in the SARS-CoV-2 cycle is still poorly understood. In this review, we examine the interactions of various coronaviruses during their infectious cycle with chaperones in search of information useful for future research on SARS-CoV-2. We also call attention to the possible role of molecular mimicry in the development of autoimmunity and its widespread pathogenic impact in COVID-19 patients. Viral proteins share highly antigenic epitopes with human chaperones, eliciting anti-viral antibodies that crossreact with the chaperones. Both, the critical functions of chaperones in the infectious cycle of viruses and the possible role of these molecules in COVID-19 autoimmune phenomena, make clear that molecular chaperones are promising candidates for the development of antiviral strategies. These could consist of inhibiting-blocking those chaperones that are necessary for the infectious viral cycle, or those that act as autoantigens in the autoimmune reactions causing generalized destructive effects on human tissues.
Keywords: COVID-19; Coronaviridae; SARS-CoV-2; chaperonopathies; chaperonotherapy; molecular chaperones; virus.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Human molecular chaperones share with SARS-CoV-2 antigenic epitopes potentially capable of eliciting autoimmunity against endothelial cells: possible role of molecular mimicry in COVID-19.Cell Stress Chaperones. 2020 Sep;25(5):737-741. doi: 10.1007/s12192-020-01148-3. Epub 2020 Aug 4. Cell Stress Chaperones. 2020. PMID: 32754823 Free PMC article.
-
Does SARS-CoV-2 Trigger Stress-InducedAutoimmunity by Molecular Mimicry? A Hypothesis.J Clin Med. 2020 Jun 29;9(7):2038. doi: 10.3390/jcm9072038. J Clin Med. 2020. PMID: 32610587 Free PMC article.
-
Effects of Cell Proteostasis Network on the Survival of SARS-CoV-2.Biol Proced Online. 2021 Feb 22;23(1):8. doi: 10.1186/s12575-021-00145-9. Biol Proced Online. 2021. PMID: 33618659 Free PMC article. Review.
-
SARS-CoV-2: From Structure to Pathology, Host Immune Response and Therapeutic Management.Microorganisms. 2020 Sep 24;8(10):1468. doi: 10.3390/microorganisms8101468. Microorganisms. 2020. PMID: 32987852 Free PMC article. Review.
-
SARS-CoV-2 and human retroelements: a case for molecular mimicry?BMC Genom Data. 2022 Apr 8;23(1):27. doi: 10.1186/s12863-022-01040-2. BMC Genom Data. 2022. PMID: 35395708 Free PMC article.
Cited by
-
Introducing Molecular Chaperones into the Causality and Prospective Management of Autoimmune Hepatitis.Dig Dis Sci. 2023 Nov;68(11):4098-4116. doi: 10.1007/s10620-023-08118-6. Epub 2023 Sep 27. Dig Dis Sci. 2023. PMID: 37755606 Free PMC article. Review.
-
Roles of antiviral sensing and type I interferon signaling in the restriction of SARS-CoV-2 replication.iScience. 2022 Jan 21;25(1):103553. doi: 10.1016/j.isci.2021.103553. Epub 2021 Dec 3. iScience. 2022. PMID: 34877479 Free PMC article.
-
Thrombotic events with or without thrombocytopenia in recipients of adenovirus-based COVID-19 vaccines.Front Cardiovasc Med. 2022 Sep 29;9:967926. doi: 10.3389/fcvm.2022.967926. eCollection 2022. Front Cardiovasc Med. 2022. PMID: 36247442 Free PMC article. Review.
-
Protein Arginylation Is Regulated during SARS-CoV-2 Infection.Viruses. 2023 Jan 19;15(2):290. doi: 10.3390/v15020290. Viruses. 2023. PMID: 36851505 Free PMC article.
-
BiP/GRP78 is a pro-viral factor for diverse dsDNA viruses that promotes the survival and proliferation of cells upon KSHV infection.PLoS Pathog. 2024 Oct 29;20(10):e1012660. doi: 10.1371/journal.ppat.1012660. eCollection 2024 Oct. PLoS Pathog. 2024. PMID: 39471213 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

