At what times during infection is SARS-CoV-2 detectable and no longer detectable using RT-PCR-based tests? A systematic review of individual participant data
- PMID: 33143712
- PMCID: PMC7609379
- DOI: 10.1186/s12916-020-01810-8
At what times during infection is SARS-CoV-2 detectable and no longer detectable using RT-PCR-based tests? A systematic review of individual participant data
Abstract
Background: Tests for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) viral ribonucleic acid (RNA) using reverse transcription polymerase chain reaction (RT-PCR) are pivotal to detecting current coronavirus disease (COVID-19) and duration of detectable virus indicating potential for infectivity.
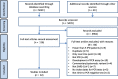
Methods: We conducted an individual participant data (IPD) systematic review of longitudinal studies of RT-PCR test results in symptomatic SARS-CoV-2. We searched PubMed, LitCOVID, medRxiv, and COVID-19 Living Evidence databases. We assessed risk of bias using a QUADAS-2 adaptation. Outcomes were the percentage of positive test results by time and the duration of detectable virus, by anatomical sampling sites.
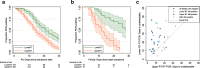
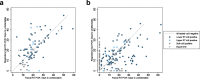
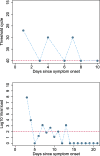
Results: Of 5078 studies screened, we included 32 studies with 1023 SARS-CoV-2 infected participants and 1619 test results, from - 6 to 66 days post-symptom onset and hospitalisation. The highest percentage virus detection was from nasopharyngeal sampling between 0 and 4 days post-symptom onset at 89% (95% confidence interval (CI) 83 to 93) dropping to 54% (95% CI 47 to 61) after 10 to 14 days. On average, duration of detectable virus was longer with lower respiratory tract (LRT) sampling than upper respiratory tract (URT). Duration of faecal and respiratory tract virus detection varied greatly within individual participants. In some participants, virus was still detectable at 46 days post-symptom onset.
Conclusions: RT-PCR misses detection of people with SARS-CoV-2 infection; early sampling minimises false negative diagnoses. Beyond 10 days post-symptom onset, lower RT or faecal testing may be preferred sampling sites. The included studies are open to substantial risk of bias, so the positivity rates are probably overestimated.
Keywords: Anatomical sampling; COVID-19; Diagnostic test; Duration virus detection; IPD; QUADAS-2; RT-PCR; SARS-CoV-2; Systematic review.
Conflict of interest statement
SM, SH, AJA, SG, KG, SAT, BS, JP, PT, PW, JS, LFR, NR, NS, JS, AW, JA, GB, KG, and BN declare no conflicts of interest for the submitted work.
CH has advised Attomarker, a spin-out company of the University of Exeter about the conduct of evaluations of its tests for COVID antibodies, but received no payment for this advice and provided it as part of academic duties at the University of Exeter.
Figures


References
-
- Sethuraman N, Jeremiah SS, Ryo A. Interpreting diagnostic tests for SARS-CoV-2. JAMA. 2020. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

