Key differences between olfactory ensheathing cells and Schwann cells regarding phagocytosis of necrotic cells: implications for transplantation therapies
- PMID: 33144615
- PMCID: PMC7642263
- DOI: 10.1038/s41598-020-75850-8
Key differences between olfactory ensheathing cells and Schwann cells regarding phagocytosis of necrotic cells: implications for transplantation therapies
Abstract
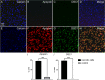
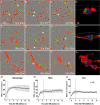
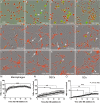
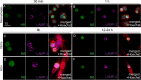
Transplantation of peripheral nervous system glia is being explored for treating neural injuries, in particular central nervous system injuries. These glia, olfactory ensheathing cells (OECs) and Schwann cells (SCs), are thought to aid regeneration by clearing necrotic cells, (necrotic bodies, NBs), as well as myelin debris. The mechanism by which the glia phagocytose and traffic NBs are not understood. Here, we show that OECs and SCs recognize phosphatidylserine on NBs, followed by engulfment and trafficking to endosomes and lysosomes. We also showed that both glia can phagocytose and process myelin debris. We compared the time-course of glial phagocytosis (of both NBs and myelin) to that of macrophages. Internalization and trafficking were considerably slower in glia than in macrophages, and OECs were more efficient phagocytes than SCs. The two glial types also differed regarding their cytokine responses after NB challenge. SCs produced low amounts of the pro-inflammatory cytokine TNF-α while OECs did not produce detectable TNF-α. Thus, OECs have a higher capacity than SCs for phagocytosis and trafficking, whilst producing lower amounts of pro-inflammatory cytokines. These findings suggest that OEC transplantation into the injured nervous system may lead to better outcomes than SC transplantation.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Phagocytosis by Peripheral Glia: Importance for Nervous System Functions and Implications in Injury and Disease.Front Cell Dev Biol. 2021 Apr 8;9:660259. doi: 10.3389/fcell.2021.660259. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33898462 Free PMC article. Review.
-
The Glia Response after Peripheral Nerve Injury: A Comparison between Schwann Cells and Olfactory Ensheathing Cells and Their Uses for Neural Regenerative Therapies.Int J Mol Sci. 2017 Jan 29;18(2):287. doi: 10.3390/ijms18020287. Int J Mol Sci. 2017. PMID: 28146061 Free PMC article. Review.
-
Transcriptional profiling predicts overwhelming homology of Schwann cells, olfactory ensheathing cells, and Schwann cell-like glia.Glia. 2014 Oct;62(10):1559-81. doi: 10.1002/glia.22700. Epub 2014 May 30. Glia. 2014. PMID: 24889922
-
Olfactory ensheathing cells exhibit unique migratory, phagocytic, and myelinating properties in the X-irradiated spinal cord not shared by Schwann cells.Glia. 2008 Nov 15;56(15):1664-78. doi: 10.1002/glia.20718. Glia. 2008. PMID: 18551623
-
Novel insights into the glia limitans of the olfactory nervous system.J Comp Neurol. 2019 May 1;527(7):1228-1244. doi: 10.1002/cne.24618. Epub 2019 Jan 16. J Comp Neurol. 2019. PMID: 30592044
Cited by
-
Designing Olfactory Ensheathing Cell Transplantation Therapies: Influence of Cell Microenvironment.Cell Transplant. 2022 Jan-Dec;31:9636897221125685. doi: 10.1177/09636897221125685. Cell Transplant. 2022. PMID: 36124646 Free PMC article. Review.
-
Neurotrophic and immunomodulatory effects of olfactory ensheathing cells as a strategy for neuroprotection and regeneration.Front Immunol. 2022 Dec 19;13:1098212. doi: 10.3389/fimmu.2022.1098212. eCollection 2022. Front Immunol. 2022. PMID: 36601122 Free PMC article. Review.
-
Improving cell transplantation by understanding and manipulating the phagocytic activity of peripheral glia.Neural Regen Res. 2022 Feb;17(2):313-314. doi: 10.4103/1673-5374.317969. Neural Regen Res. 2022. PMID: 34269197 Free PMC article. No abstract available.
-
Olfactory ensheathing cells and neuropathic pain.Front Cell Dev Biol. 2023 Apr 5;11:1147242. doi: 10.3389/fcell.2023.1147242. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37223000 Free PMC article. Review.
-
Olfactory Dysfunction in a Novel Model of Prodromal Parkinson's Disease in Adult Zebrafish.Int J Mol Sci. 2025 May 8;26(10):4474. doi: 10.3390/ijms26104474. Int J Mol Sci. 2025. PMID: 40429620 Free PMC article.
References
-
- Magnus T, Chan A, Linker RA, Toyka KV, Gold R. Astrocytes are less efficient in the removal of apoptotic lymphocytes than microglia cells: Implications for the role of glial cells in the inflamed central nervous system. J. Neuropathol. Exp. Neurol. 2002;61:760–766. doi: 10.1093/jnen/61.9.760. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

