Chemically Defined, High-Density Insect Cell-Based Expression System for Scalable AAV Vector Production
- PMID: 33145369
- PMCID: PMC7591331
- DOI: 10.1016/j.omtm.2020.09.018
Chemically Defined, High-Density Insect Cell-Based Expression System for Scalable AAV Vector Production
Abstract
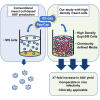
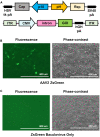
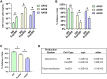
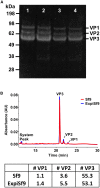
The recombinant adeno-associated virus (AAV) vector is one of the most utilized viral vectors in gene therapy due to its robust, long-term in vivo transgene expression and low toxicity. One major hurdle for clinical AAV applications is large-scale manufacturing. In this regard, the baculovirus-based AAV production system is highly attractive due to its scalability and predictable biosafety. Here, we describe a simple method to improve the baculovirus-based AAV production using the ExpiSf Baculovirus Expression System with a chemically defined medium for suspension culture of high-density ExpiSf9 cells. Baculovirus-infected ExpiSf9 cells produced up to 5 × 1011 genome copies of highly purified AAV vectors per 1 mL of suspension culture, which is up to a 19-fold higher yield than the titers we obtained from the conventional Sf9 cell-based system. When mice were administered the same dose of AAV vectors, we saw comparable transduction efficiency and biodistributions between the vectors made in ExpiSf9 and Sf9 cells. Thus, the ExpiSf Baculovirus Expression System would support facile and scalable AAV manufacturing amenable for preclinical and clinical applications.
Keywords: AAV; baculovirus; insect cell.
© 2020 The Authors.
Figures

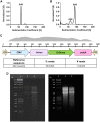
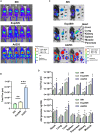
References
-
- U.S. Food & Drug Administration LUXTURNA. 2018. https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-produ...
-
- U.S. Food & Drug Administration ZOLGENSMA. 2020. https://www.fda.gov/vaccines-blood-biologics/zolgensma
-
- Matsushita T., Elliger S., Elliger C., Podsakoff G., Villarreal L., Kurtzman G.J., Iwaki Y., Colosi P. Adeno-associated virus vectors can be efficiently produced without helper virus. Gene Ther. 1998;5:938–945. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

