Rapamycin-mediated mouse lifespan extension: Late-life dosage regimes with sex-specific effects
- PMID: 33145977
- PMCID: PMC7681050
- DOI: 10.1111/acel.13269
Rapamycin-mediated mouse lifespan extension: Late-life dosage regimes with sex-specific effects
Abstract
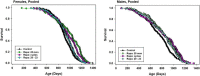
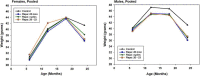
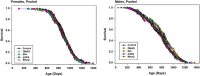
To see if variations in timing of rapamycin (Rapa), administered to middle aged mice starting at 20 months, would lead to different survival outcomes, we compared three dosing regimens. Initiation of Rapa at 42 ppm increased survival significantly in both male and female mice. Exposure to Rapa for a 3-month period led to significant longevity benefit in males only. Protocols in which each month of Rapa treatment was followed by a month without Rapa exposure were also effective in both sexes, though this approach was less effective than continuous exposure in female mice. Interpretation of these results is made more complicated by unanticipated variation in patterns of weight gain, prior to the initiation of the Rapa treatment, presumably due to the use of drug-free food from two different suppliers. The experimental design included tests of four other drugs, minocycline, β-guanidinopropionic acid, MitoQ, and 17-dimethylaminoethylamino-17-demethoxygeldanamycin (17-DMAG), but none of these led to a change in survival in either sex.
Keywords: 17-DMAG; MitoQ; minocycline; rapamycin; survival; β-GPA.
© 2020 The Authors. Aging Cell published by the Anatomical Society and John Wiley & Sons Ltd.
Conflict of interest statement
The University of Texas Health Science Center at San Antonio has applied for a patent, U.S. Patent Application No. 13/128,800, by inventors Zelton Dave Sharp and Randy Strong, for an encapsulated rapamycin formulation used in this paper. Under a licensing agreement between Emtora Biosciences (formerly Rapamycin Holdings, Inc.) and the University of Texas Health Science Center San Antonio, R. Strong, and Z.D. Sharp, the University is entitled to milestone payments and royalty on sales of microencapsulated rapamycin. The university has a plan for managing conflicts of interest under its “Policy and Procedures for Promoting Objectivity in Research by Managing, Reducing or Eliminating Conflicts of Interest.” Michael P. Murphy consults for Antipodean Pharmaceuticals Inc., which is developing MitoQ as a potential therapy and also holds patents in the use of MitoQ.
Figures
References
-
- Adlam, V. J. , Harrison, J. C. , Porteous, C. M. , James, A. M. , Smith, R. A. J. , Murphy, M. P. , & Sammut, I. A. (2005). Targeting an antioxidant to mitochondria decreases cardiac ischemia‐reperfusion injury. The FASEB Journal, 19, 1088–1095. - PubMed
-
- Anisimov, V. N. , Zabezhinski, M. A. , Popovich, I. G. , Piskunova, T. S. , Semenchenko, A. V. , Tyndyk, M. L. , Yurova, M. N. , Rosenfeld, S. V. , & Blagosklonny, M. V. (2011). Rapamyin increases lifespan and inhibits spontaneous tumorigenesis in inbred female mice. Cell Cycle, 10, 4230–4236. - PubMed
-
- Arriola Apelo, S. I. , Neuman, J. C. , Baar, E. L. , Syed, F. A. , Cummings, N. E. , Brar, H. K. , Pumper, C. P. , Kimple, M. E. , & Lamming, D. W. (2016). Alternative rapamycin treatment regimens mitigate the impact of rapamycin on glucose homeostasis and the immune system. Aging Cell, 15, 28–33. - PMC - PubMed
-
- Bergeron, R. , Ren, J. M. , Cadman, K. S. , Moore, I. K. , Perret, P. , Pypaert, M. , Young, L. H. , Semenkovich, C. F. , & Shulman, G. I. (2001). Chronic activation of AMP kinase results in NRF‐1 activation and mitochondrial biogenesis. American Journal of Physiology – Endocrinology and Metabolism, 281, E1340–E1346. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

