Safety and cost-effectiveness of individualised screening for diabetic retinopathy: the ISDR open-label, equivalence RCT
- PMID: 33146763
- PMCID: PMC7716929
- DOI: 10.1007/s00125-020-05313-2
Safety and cost-effectiveness of individualised screening for diabetic retinopathy: the ISDR open-label, equivalence RCT
Abstract
Aims/hypothesis: Using variable diabetic retinopathy screening intervals, informed by personal risk levels, offers improved engagement of people with diabetes and reallocation of resources to high-risk groups, while addressing the increasing prevalence of diabetes. However, safety data on extending screening intervals are minimal. The aim of this study was to evaluate the safety and cost-effectiveness of individualised, variable-interval, risk-based population screening compared with usual care, with wide-ranging input from individuals with diabetes.
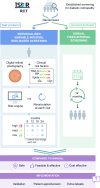
Methods: This was a two-arm, parallel-assignment, equivalence RCT (minimum 2 year follow-up) in individuals with diabetes aged 12 years or older registered with a single English screening programme. Participants were randomly allocated 1:1 at baseline to individualised screening at 6, 12 or 24 months for those at high, medium and low risk, respectively, as determined at each screening episode by a risk-calculation engine using local demographic, screening and clinical data, or to annual screening (control group). Screening staff and investigators were observer-masked to allocation and interval. Data were collected within the screening programme. The primary outcome was attendance (safety). A secondary safety outcome was the development of sight-threatening diabetic retinopathy. Cost-effectiveness was evaluated within a 2 year time horizon from National Health Service and societal perspectives.
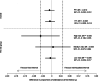
Results: A total of 4534 participants were randomised. After withdrawals, there were 2097 participants in the individualised screening arm and 2224 in the control arm. Attendance rates at first follow-up were equivalent between the two arms (individualised screening 83.6%; control arm 84.7%; difference -1.0 [95% CI -3.2, 1.2]), while sight-threatening diabetic retinopathy detection rates were non-inferior in the individualised screening arm (individualised screening 1.4%, control arm 1.7%; difference -0.3 [95% CI -1.1, 0.5]). Sensitivity analyses confirmed these findings. No important adverse events were observed. Mean differences in complete case quality-adjusted life-years (EuroQol Five-Dimension Questionnaire, Health Utilities Index Mark 3) did not significantly differ from zero; multiple imputation supported the dominance of individualised screening. Incremental cost savings per person with individualised screening were £17.34 (95% CI 17.02, 17.67) from the National Health Service perspective and £23.11 (95% CI 22.73, 23.53) from the societal perspective, representing a 21% reduction in overall programme costs. Overall, 43.2% fewer screening appointments were required in the individualised arm.
Conclusions/interpretation: Stakeholders involved in diabetes care can be reassured by this study, which is the largest ophthalmic RCT in diabetic retinopathy screening to date, that extended and individualised, variable-interval, risk-based screening is feasible and can be safely and cost-effectively introduced in established systematic programmes. Because of the 2 year time horizon of the trial and the long time frame of the disease, robust monitoring of attendance and retinopathy rates should be included in any future implementation.
Trial registration: ISRCTN 87561257 FUNDING: The study was funded by the UK National Institute for Health Research. Graphical abstract.
Keywords: Diabetic retinopathy; Individualised; Personalised; Risk-based; Screening; Systematic; Variable interval.
Figures


References
-
- The Saint Vincent Declaration on diabetes care and research in Europe Acta Diabetol. 1989;10(Suppl):143–144.
-
- International Diabetes Federation (2019) IDF diabetes atlas, 9th edn. International Diabetes Federation. Available from www.diabetesatlas.org. Accessed 1 Oct 2020
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

