PTP-3 phosphatase promotes intramolecular folding of SYD-2 to inactivate kinesin-3 UNC-104 in neurons
- PMID: 33147118
- PMCID: PMC7927192
- DOI: 10.1091/mbc.E19-10-0591
PTP-3 phosphatase promotes intramolecular folding of SYD-2 to inactivate kinesin-3 UNC-104 in neurons
Abstract
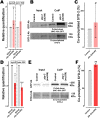
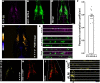
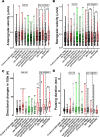
UNC-104 is the Caenorhabditis elegans homolog of kinesin-3 KIF1A known for its fast shuffling of synaptic vesicle protein transport vesicles in axons. SYD-2 is the homolog of liprin-α in C. elegans known to activate UNC-104; however, signals that trigger SYD-2 binding to the motor remain unknown. Because SYD-2 is a substrate of PTP-3/LAR PTPR, we speculate a role of this phosphatase in SYD-2-mediated motor activation. Indeed, coimmunoprecipitation assays revealed increased interaction between UNC-104 and SYD-2 in ptp-3 knockout worms. Intramolecular FRET analysis in living nematodes demonstrates that SYD-2 largely exists in an open conformation state in ptp-3 mutants. These assays also revealed that nonphosphorylatable SYD-2 (Y741F) exists predominately in folded conformations, while phosphomimicking SYD-2 (Y741E) primarily exists in open conformations. Increased UNC-104 motor clustering was observed along axons likely as a result of elevated SYD-2 scaffolding function in ptp-3 mutants. Also, both motor velocities as well as cargo transport speeds were visibly increased in neurons of ptp-3 mutants. Lastly, epistatic analysis revealed that PTP-3 is upstream of SYD-2 to regulate its intramolecular folding.
Figures




Similar articles
-
LRK-1/LRRK2 and AP-3 regulate trafficking of synaptic vesicle precursors through active zone protein SYD-2/Liprin-α.PLoS Genet. 2024 May 9;20(5):e1011253. doi: 10.1371/journal.pgen.1011253. eCollection 2024 May. PLoS Genet. 2024. PMID: 38722918 Free PMC article.
-
Synaptic scaffolding protein SYD-2 clusters and activates kinesin-3 UNC-104 in C. elegans.Proc Natl Acad Sci U S A. 2009 Nov 17;106(46):19605-10. doi: 10.1073/pnas.0902949106. Epub 2009 Oct 30. Proc Natl Acad Sci U S A. 2009. PMID: 19880746 Free PMC article.
-
Identification and Characterization of LIN-2(CASK) as a Regulator of Kinesin-3 UNC-104(KIF1A) Motility and Clustering in Neurons.Traffic. 2016 Aug;17(8):891-907. doi: 10.1111/tra.12413. Epub 2016 Jun 3. Traffic. 2016. PMID: 27172328
-
Sub-cellular distribution of UNC-104(KIF1A) upon binding to adaptors as UNC-16(JIP3), DNC-1(DCTN1/Glued) and SYD-2(Liprin-α) in C. elegans neurons.Neuroscience. 2011 Mar 10;176:39-52. doi: 10.1016/j.neuroscience.2010.12.044. Epub 2010 Dec 30. Neuroscience. 2011. PMID: 21195138
-
SYD-1 Promotes Multiple Developmental Steps Leading to Neuronal Connectivity.Mol Neurobiol. 2016 Dec;53(10):6768-6773. doi: 10.1007/s12035-015-9592-5. Epub 2015 Dec 11. Mol Neurobiol. 2016. PMID: 26660112 Free PMC article. Review.
Cited by
-
LRK-1/LRRK2 and AP-3 regulate trafficking of synaptic vesicle precursors through active zone protein SYD-2/Liprin-α.PLoS Genet. 2024 May 9;20(5):e1011253. doi: 10.1371/journal.pgen.1011253. eCollection 2024 May. PLoS Genet. 2024. PMID: 38722918 Free PMC article.
References
-
- Ackley BD, Harrington RJ, Hudson ML, Williams L, Kenyon CJ, Chisholm AD, Jin Y (2005). The two isoforms of the Caenorhabditis elegans leukocyte-common antigen related receptor tyrosine phosphatase PTP-3 function independently in axon guidance and synapse formation. J Neurosci 25, 7517–7528. - PMC - PubMed
-
- Blangy A, Lane HA, d’Herin P, Harper M, Kress M, Nigg EA (1995). Phosphorylation by p34cdc2 regulates spindle association of human Eg5, a kinesin-related motor essential for bipolar spindle formation in vivo. Cell 83, 1159–1169. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

