Allyl Cyanate/Isocyanate Rearrangement in Glycals: Stereoselective Synthesis of 1-Amino and Diamino Sugar Derivatives
- PMID: 33147974
- PMCID: PMC7735751
- DOI: 10.1021/acs.orglett.0c03438
Allyl Cyanate/Isocyanate Rearrangement in Glycals: Stereoselective Synthesis of 1-Amino and Diamino Sugar Derivatives
Abstract
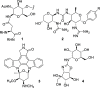
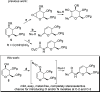
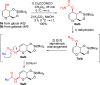
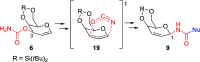
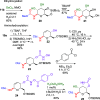
The [3,3]-sigmatropic allyl cyanate/isocyanate rearrangement of glycals in the presence of O-, N-, and C-nucleophiles afforded β-N-glucosyl and galactosyl carbamates, ureas, and amides in good yields. The unsaturated products were elaborated to N-glycosides by dihydroxylation, to 1,3-diaminosugars by tethered aminohydroxylation, or to 1,2-diaminosugars by iteration of the sigmatropic rearrangement. This metal-free methodology represents an excellent and general method for the stereoselective synthesis of N-glycosides and diamino sugars with complete transmission of stereochemical information.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

References
-
- Horton D.; Wander J. D. In The Carbohydrates; Pigman W., Horton D., Eds.; Academic Press: New York, 1980;, Vol. 1B, pp 643–760.
- Banaszek A.; Mlynarski J.. Recent advances in the chemistry of bioactive 3-deoxy-ulosonic acids. In Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Elsevier, Amsterdam; 2005; Vol. 30, p 419–482.
- Gilbert D. N.Aminoglycosides. In Principles and Practice of Infectious Diseases, 6th ed.; Mandell G. L., Bennett J. E., Dolin R., Eds.; Churchill Livingstone: New York, 2005; pp 328–356.
-
For selected reviews on the synthesis of aminosugars, see:
- Witczak Z. J.; Bielski R. Recent Examples of Novel Synthetic Approaches to Diverse Amino Sugars. Carbohydr. Chem. 2016, 42, 344–367. 10.1039/9781782626657-00344. - DOI
- Pfrengle F.; Reissig H.-U. Amino Sugars and their Mimetics via 1,2-Oxazines. Chem. Soc. Rev. 2010, 39, 549–557. 10.1039/B914356D. - DOI - PubMed
- Skarbek K.; Milewska M. J. Biosynthetic and Synthetic Access to Amino Sugars. Carbohydr. Res. 2016, 434, 44–71. 10.1016/j.carres.2016.08.005. - DOI - PubMed
-
- Dwek R. A. Glycobiology: Toward Understanding the Function of Sugars. Chem. Rev. 1996, 96, 683–720. 10.1021/cr940283b. - DOI - PubMed
- Nicolas C.; Martin O. R. A Practical Approach to Dideoxy-1,4- and 1,5-Iminopentitols from Protected Sugar Hemiacetals. Molecules 2018, 23, 1612–1641. 10.3390/molecules23071612. - DOI - PubMed
-
- Bruno S.; Ardelt B.; Skierski J. S.; Traganos F.; Darzynkiewicz Z. Different Effects of Staurosporine, an Inhibitor of Protein Kinases, on the Cell Cycle and Chromatin Structure of Normal and Leukemic Lymphocytes. Cancer Res. 1992, 52, 470–473. - PubMed
LinkOut - more resources
Full Text Sources

