Lipotoxicity, Nutrient-Sensing Signals, and Autophagy in Diabetic Nephropathy
- PMID: 33150239
- PMCID: PMC7590395
- DOI: 10.31662/jmaj.2020-0005
Lipotoxicity, Nutrient-Sensing Signals, and Autophagy in Diabetic Nephropathy
Abstract
Diabetic nephropathy is a leading cause of proteinuria, kidney fibrosis, and subsequent end-stage renal disease. The renal prognosis of diabetic patients with refractory proteinuria is extremely poor. Therefore, identification of novel therapeutic targets to combat this serious condition and improve renal prognosis is urgently necessary. In diabetic patients, in addition to blood glucose levels, serum levels of free fatty acids (FFAs) are chronically elevated, even during postprandial periods. Of the various types of FFAs, saturated FFAs are highly cytotoxic and their levels are elevated in the serum of patients with diabetes. Thus, an increase in saturated FFAs is currently thought to contribute to proximal tubular cell damage and podocyte injury in diabetic nephropathy. Therefore, protecting both types of kidney cells from saturated FFA-related lipotoxicity may become a novel therapeutic approach for diabetic patients with refractory proteinuria. Interestingly, accumulating evidence suggests that controlling intracellular nutrient signals and autophagy can ameliorate the FFA-related kidney damage. Here, we review the evidence indicating possible mechanisms underlying cell injury caused by saturated FFAs and cell protective roles of intracellular nutrient signals and autophagy in diabetic nephropathy.
Keywords: Free fatty acid; autophagy; lipotoxicity; podocyte; proteinuria; proximal tubular cell.
Copyright © Japan Medical Association.
Conflict of interest statement
None
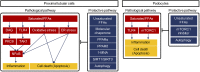
Figures

References
-
- UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352(9131):837-53. - PubMed
-
- The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329(14):977-86. - PubMed
-
- Ohkubo Y, Kishikawa H, Araki E, et al. Intensive insulin therapy prevents the progression of diabetic microvascular complications in Japanese patients with non-insulin-dependent diabetes mellitus: a randomized prospective 6-year study. Diabetes Res Clin Pract. 1995;28(2):103-17. - PubMed
-
- Brenner BM, Cooper ME, de Zeeuw D, et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345(12):861-9. - PubMed
-
- Isshiki K, Haneda M, Koya D, et al. Thiazolidinedione compounds ameliorate glomerular dysfunction independent of their insulin-sensitizing action in diabetic rats. Diabetes. 2000;49(6):1022-32. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
