Evaluation of serum high-mobility group box 1 concentration in dogs with epilepsy: A case-control study
- PMID: 33150666
- PMCID: PMC7694863
- DOI: 10.1111/jvim.15940
Evaluation of serum high-mobility group box 1 concentration in dogs with epilepsy: A case-control study
Abstract
Background: High-mobility group box 1 (HMGB1) is a key mediator of neuroinflammation and there are increased HMGB1 levels in laboratory animal models of epilepsy and human patients with epilepsy.
Objectives: To determine serum HMGB1 levels in dogs with epilepsy.
Animals: Twenty-eight epileptic dogs, 12 dogs with nonepileptic brain diseases, and 26 healthy dogs.
Methods: In this case-control study, serum HMGB1 concentrations were estimated using the canine-specific enzyme-linked immunosorbent assay kit. Diagnosis of dogs with epilepsy was based on medical history, physical and neurological examination findings, laboratory test results, magnetic resonance image, and cerebrospinal fluid analysis.
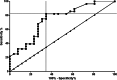
Results: Serum HMGB1 levels were significantly higher in epileptic dogs (median = 0.41 ng/mL; range, 0.03-5.28) than in healthy dogs (median = 0.12 ng/mL; range, 0.02-1.45; P = .002). In contrast, serum HMGB1 levels of dogs with non-epileptic brain diseases (median = 0.19 ng/mL; range, 0.03-1.04) were not significantly increased compared to those of healthy dogs (P = .12). Regarding idiopathic epilepsy, dogs with an epilepsy course of >3 months showed a higher serum HMGB1 concentration (median = 0.87 ng/mL; range, 0.42-2.88) than those with that of ≤3 months (median = 0.26 ng/mL; range, 0.03-0.88; P = .02).
Conclusions and clinical importance: Serum HMGB1 could be a biomarker of epilepsy.
Keywords: HMGB1; biomarker; canine; seizure.
© 2020 The Authors. Journal of Veterinary Internal Medicine published by Wiley Periodicals LLC on behalf of American College of Veterinary Internal Medicine.
Conflict of interest statement
Authors declare no conflict of interest.
Figures




Similar articles
-
Evaluation of serum C-reactive protein and high mobility group box 1 concentrations in 22 dogs with acute pancreatitis: a pilot study.Vet Q. 2019 Dec;39(1):122-130. doi: 10.1080/01652176.2019.1655178. Vet Q. 2019. PMID: 31401946 Free PMC article.
-
High mobility group box 1 (HMGB1) protein is present in the cerebrospinal fluid of dogs with encephalitis.J Vet Med Sci. 2011 Jul;73(7):917-22. doi: 10.1292/jvms.10-0444. Epub 2011 Mar 25. J Vet Med Sci. 2011. PMID: 21441719
-
Clinical signs, duodenal histopathological grades, and serum high-mobility group box 1 concentrations in dogs with inflammatory bowel disease.J Vet Intern Med. 2021 Sep;35(5):2205-2214. doi: 10.1111/jvim.16258. Epub 2021 Sep 4. J Vet Intern Med. 2021. PMID: 34480505 Free PMC article.
-
Meta-analysis of HMGB1 levels in the cerebrospinal fluid and serum of patients with epilepsy.Neurol Sci. 2023 Jul;44(7):2329-2337. doi: 10.1007/s10072-023-06720-0. Epub 2023 Mar 18. Neurol Sci. 2023. PMID: 36933099 Review.
-
High Mobility Group Box 1 is a novel pathogenic factor and a mechanistic biomarker for epilepsy.Brain Behav Immun. 2018 Aug;72:14-21. doi: 10.1016/j.bbi.2017.10.008. Epub 2017 Oct 13. Brain Behav Immun. 2018. PMID: 29031614 Review.
Cited by
-
Dogs as a Natural Animal Model of Epilepsy.Front Vet Sci. 2022 Jun 22;9:928009. doi: 10.3389/fvets.2022.928009. eCollection 2022. Front Vet Sci. 2022. PMID: 35812852 Free PMC article. Review.
-
Evaluation of the covariation between leukotriene B4, prostaglandin E2, and hematologic inflammatory parameters in a canine pentylenetetrazole-induced seizure model.Front Neurosci. 2024 Dec 11;18:1451902. doi: 10.3389/fnins.2024.1451902. eCollection 2024. Front Neurosci. 2024. PMID: 39723425 Free PMC article.
-
Serum concentrations of complement C3 and C4 in dogs with idiopathic epilepsy.J Vet Intern Med. 2024 Mar-Apr;38(2):1074-1082. doi: 10.1111/jvim.17008. Epub 2024 Feb 8. J Vet Intern Med. 2024. PMID: 38329151 Free PMC article.
References
-
- Vezzani A, Granata T. Brain inflammation in epilepsy: experimental and clinical evidence. Epilepsia. 2005;46:1‐20. - PubMed
-
- van Vliet EA, Aronica E, Vezzani A, Ravizza T. Review: neuroinflammatory pathways as treatment targets and biomarker candidates in epilepsy: emerging evidence from preclinical and clinical studies. Neuropathol Appl Neurobiol. 2018;44:91‐111. - PubMed

