LncRNA-modulated autophagy in plaque cells: a new paradigm of gene regulation in atherosclerosis?
- PMID: 33154191
- PMCID: PMC7695379
- DOI: 10.18632/aging.103786
LncRNA-modulated autophagy in plaque cells: a new paradigm of gene regulation in atherosclerosis?
Abstract
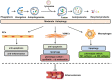
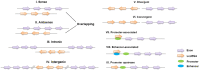
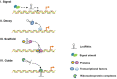
The development of atherosclerosis is accompanied by the functional deterioration of plaque cells, which leads to the escalation of endothelial inflammation, abnormal vascular smooth muscle cell phenotype switching and the accumulation of lipid-laden macrophages within vascular walls. Autophagy, a highly conserved homeostatic mechanism, is critical for the delivery of cytoplasmic substrates to lysosomes for degradation. Moderate levels of autophagy prevent atherosclerosis by safeguarding plaque cells against apoptosis, preventing inflammation, and limiting the lipid burden, whereas excessive autophagy exacerbates cell damage and inflammation and thereby accelerates the formation of atherosclerotic plaques. Increasing lines of evidence suggest that long noncoding RNAs can be either beneficial or detrimental to atherosclerosis development by regulating the autophagy level. This review summarizes the research progress related to 1) the significant role of autophagy in atherosclerosis and 2) the effects of the lncRNA-mediated modulation of autophagy on the plaque cell fate, inflammation levels, proliferative capacity, and cholesterol metabolism and subsequently on atherogenesis.
Keywords: atherosclerosis; autophagy; long noncoding RNAs.
Conflict of interest statement
Figures
Similar articles
-
Emerging Paradigms in Atherosclerosis: The Impact of LncRNA PSMB8-AS1 on Vascular Inflammation and Plaque Formation.J Cardiovasc Transl Res. 2024 Aug;17(4):779-781. doi: 10.1007/s12265-024-10481-2. Epub 2024 Jan 19. J Cardiovasc Transl Res. 2024. PMID: 38240920 No abstract available.
-
Non-coding RNA regulation of endothelial and macrophage functions during atherosclerosis.Vascul Pharmacol. 2019 Mar;114:64-75. doi: 10.1016/j.vph.2018.03.001. Epub 2018 Mar 15. Vascul Pharmacol. 2019. PMID: 29551552 Free PMC article. Review.
-
Long Noncoding RNA-CERNA1 Stabilized Atherosclerotic Plaques in apolipoprotein E-/- Mice.J Cardiovasc Transl Res. 2019 Oct;12(5):425-434. doi: 10.1007/s12265-019-09883-4. Epub 2019 Mar 19. J Cardiovasc Transl Res. 2019. PMID: 30888631
-
TUG1 knockdown ameliorates atherosclerosis via up-regulating the expression of miR-133a target gene FGF1.Cardiovasc Pathol. 2018 Mar-Apr;33:6-15. doi: 10.1016/j.carpath.2017.11.004. Epub 2017 Dec 2. Cardiovasc Pathol. 2018. PMID: 29268138
-
Autophagy and Its Association with Macrophages in Clonal Hematopoiesis Leading to Atherosclerosis.Int J Mol Sci. 2025 Apr 1;26(7):3252. doi: 10.3390/ijms26073252. Int J Mol Sci. 2025. PMID: 40244103 Free PMC article. Review.
Cited by
-
Laminar shear stress inhibits inflammation by activating autophagy in human aortic endothelial cells through HMGB1 nuclear translocation.Commun Biol. 2022 May 6;5(1):425. doi: 10.1038/s42003-022-03392-y. Commun Biol. 2022. PMID: 35523945 Free PMC article.
-
A bibliometric analysis of autophagy in atherosclerosis from 2012 to 2021.Front Pharmacol. 2022 Sep 15;13:977870. doi: 10.3389/fphar.2022.977870. eCollection 2022. Front Pharmacol. 2022. PMID: 36188570 Free PMC article.
-
Role of advanced glycation end products on vascular smooth muscle cells under diabetic atherosclerosis.Front Endocrinol (Lausanne). 2022 Aug 31;13:983723. doi: 10.3389/fendo.2022.983723. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 36120471 Free PMC article. Review.
-
Two sides of the same coin: Non-alcoholic fatty liver disease and atherosclerosis.Vascul Pharmacol. 2024 Mar;154:107249. doi: 10.1016/j.vph.2023.107249. Epub 2023 Dec 7. Vascul Pharmacol. 2024. PMID: 38070759 Free PMC article. Review.
References
-
- Collura S, Morsiani C, Vacirca A, Fronterrè S, Ciavarella C, Vasuri F, D’Errico A, Franceschi C, Pasquinelli G, Gargiulo M, Capri M. The carotid plaque as paradigmatic case of site-specific acceleration of aging process: the microRNAs and the inflammaging contribution. Ageing Res Rev. 2020; 61:101090. 10.1016/j.arr.2020.101090 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

