The design of a Bayesian platform trial to prevent and eradicate inhibitors in patients with hemophilia
- PMID: 33156923
- PMCID: PMC7656929
- DOI: 10.1182/bloodadvances.2020002789
The design of a Bayesian platform trial to prevent and eradicate inhibitors in patients with hemophilia
Abstract
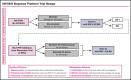
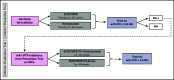
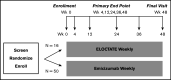
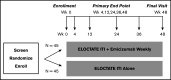
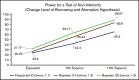
Among individuals with the rare congenital bleeding disorder hemophilia A, the major challenge is inhibitor formation, which is associated with significant morbidity and cost. Yet, as the optimal approach to prevent and eradicate inhibitors is not known, we are at equipoise. Because classic trial design is not practical in a rare disease setting, we designed 2 48-week randomized trials comparing ELOCTATE and emicizumab to prevent and eradicate inhibitors. To achieve statistical efficiency, we incorporated historic data (Bayesian priors) on inhibitor formation to allow preferential randomization to emicizumab, piecewise exponential survival models to determine mean and 95% confidence interval for inhibitor formation in each arm, and simulations to determine the best model design to optimize power. To achieve administrative efficiency, the trials will be performed with the same sites, staff, visit frequency, blood sampling, laboratories, and laboratory assays, with streamlined enrollment so patients developing inhibitors in the first trial may be enrolled on the second trial. The primary end point is the probability of inhibitor formation or inhibitor eradication, respectively. The design indicates early stopping rules for overwhelming evidence of superiority of the emicizumab arms. Simulations indicate that, with 66 subjects, the Prevention Trial will have 84% power to detect noninferiority of emicizumab to ELOCTATE with a margin of 10% if emicizumab is truly 10% superior to ELOCTATE; with 90 subjects, the Eradication Trial will have 80% power to detect 15% superiority of ELOCTATE immune tolerance induction with vs without emicizumab. Thus, a platform design provides statistical and administrative efficiency to conduct INHIBIT trials.
© 2020 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: M.M.B. is a member of a Data and Safety Monitoring Board for Cerus Corporation. M.V.R. has received research funding from Alnylam Pharmaceuticals, BioMarin, Bioverativ, Sangamo Therapeutics, Spark Therapeutics, and Takeda Pharmaceutical and has served on advisory boards for Alnylam Pharmaceuticals, BioMarin, Bioverativ, and Spark Therapeutics. M.B. declares no competing financial interests.
Figures
References
-
- Iorio A, Halimeh S, Holzhauer S, et al. . Rate of inhibitor development in previously untreated hemophilia A patients treated with plasma-derived or recombinant factor VIII concentrates: a systematic review. J Thromb Haemost. 2010;8(6):1256-1265. - PubMed
-
- Gouw van den Berg, SC HM, Fischer K, et al. Intensity of factor VIII treatment and inhibitor development in children with severe hemophilia A: the RODIN study. Blood. 2013;121(20):4046-4055. - PubMed
-
- Soucie JM, Symons J IV, Evatt B, Brettler D, Huszti H, Linden J; Hemophilia Surveillance System Project Investigators . Home-based factor infusion therapy and hospitalization for bleeding complications among males with haemophilia. Haemophilia. 2001;7(2):198-206. - PubMed
-
- Goudemand J. Pharmaco-economic aspects of inhibitor treatment. Eur J Haematol Suppl. 1998;63:24-27. - PubMed

