The therapeutic value of XL388 in human glioma cells
- PMID: 33159013
- PMCID: PMC7746352
- DOI: 10.18632/aging.103791
The therapeutic value of XL388 in human glioma cells
Abstract
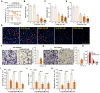
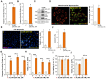
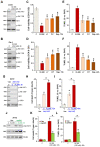
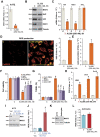
XL388 is a highly efficient and orally-available ATP-competitive PI3K-mTOR dual inhibitor. Its activity against glioma cells was studied here. In established and primary human glioma cells, XL388 potently inhibited cell survival and proliferation as well as cell migration, invasion and cell cycle progression. The dual inhibitor induced significant apoptosis activation in glioma cells. In A172 cells and primary human glioma cells, XL388 inhibited Akt-mTORC1/2 activation by blocking phosphorylation of Akt and S6K1. XL388-induced glioma cell death was only partially attenuated by a constitutively-active mutant Akt1. Furthermore, it was cytotoxic against Akt1-knockout A172 glioma cells. XL388 downregulated MAF bZIP transcription factor G (MAFG) and inhibited Nrf2 signaling, causing oxidative injury in glioma cells. Conversely, antioxidants, n-acetylcysteine, pyrrolidine dithiocarbamate and AGI-106, alleviated XL388-induced cytotoxicity and apoptosis in glioma cells. Oral administration of XL388 inhibited subcutaneous A172 xenograft growth in severe combined immunodeficient mice. Akt-S6K1 inhibition and MAFG downregulation were detected in XL388-treated A172 xenograft tissues. Collectively, XL388 efficiently inhibits human glioma cell growth, through Akt-mTOR-dependent and -independent mechanisms.
Keywords: Akt; MAFG; XL388; glioma; mTOR.
Conflict of interest statement
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

