Chest CT in the emergency department for suspected COVID-19 pneumonia
- PMID: 33165767
- PMCID: PMC7649305
- DOI: 10.1007/s11547-020-01302-y
Chest CT in the emergency department for suspected COVID-19 pneumonia
Abstract
Purpose: In overwhelmed emergency departments (EDs) facing COVID-19 outbreak, a swift diagnosis is imperative. CT role was widely debated for its limited specificity. Here we report the diagnostic role of CT in two EDs in Lombardy, epicenter of Italian outbreak.
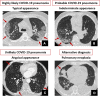
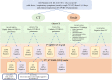
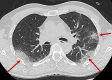
Material and methods: Admitting chest CT from 142 consecutive patients with suspected COVID-19 were retrospectively analyzed. CT scans were classified in "highly likely," "likely," and "unlikely" COVID-19 pneumonia according to the presence of typical, indeterminate, and atypical findings, or "negative" in the absence of findings, or "alternative diagnosis" when a different diagnosis was found. Nasopharyngeal swab results, turnaround time, and time to positive results were collected. CT diagnostic performances were assessed considering RT-PCR as reference standard.
Results: Most of cases (96/142, 68%) were classified as "highly likely" COVID-19 pneumonia. Ten (7%) and seven (5%) patients were classified as "likely" and "unlikely" COVID-19 pneumonia, respectively. In 21 (15%) patients a differential diagnosis was provided, including typical pneumonia, pulmonary edema, neoplasia, and pulmonary embolism. CT was negative in 8/142 (6%) patients. Mean turnaround time for the first COVID-19 RT-PCR was 30 ± 13 h. CT diagnostic accuracy in respect of the first test swab was 79% and increased to 91.5% after repeated swabs and/or BAL, for 18 false-negative first swab. CT performance was good with 76% specificity, 99% sensitivity, 90% positive predictive value and 97% negative predictive value.
Conclusion: Chest CT was useful to streamline patients' triage while waiting for RT-PCR in the ED, supporting the clinical suspicion of COVID-19 or providing alternative diagnosis.
Keywords: COVID-19; Computed tomography; Diagnosis; Emergency; Triage.
Conflict of interest statement
Authors have no conflict of interest to disclose.
Figures
Similar articles
-
Unenhanced computed tomography (CT) utility for triage at the emergency department during COVID-19 pandemic.Am J Emerg Med. 2021 Aug;46:260-265. doi: 10.1016/j.ajem.2020.07.058. Epub 2020 Jul 28. Am J Emerg Med. 2021. PMID: 33046310 Free PMC article.
-
Chest CT for rapid triage of patients in multiple emergency departments during COVID-19 epidemic: experience report from a large French university hospital.Eur Radiol. 2021 Feb;31(2):795-803. doi: 10.1007/s00330-020-07154-4. Epub 2020 Aug 19. Eur Radiol. 2021. PMID: 32813105 Free PMC article.
-
Chest CT in patients with a moderate or high pretest probability of COVID-19 and negative swab.Radiol Med. 2020 Dec;125(12):1260-1270. doi: 10.1007/s11547-020-01269-w. Epub 2020 Aug 29. Radiol Med. 2020. PMID: 32862406 Free PMC article.
-
Thoracic imaging tests for the diagnosis of COVID-19.Cochrane Database Syst Rev. 2020 Nov 26;11:CD013639. doi: 10.1002/14651858.CD013639.pub3. Cochrane Database Syst Rev. 2020. Update in: Cochrane Database Syst Rev. 2021 Mar 16;3:CD013639. doi: 10.1002/14651858.CD013639.pub4. PMID: 33242342 Updated.
-
Thoracic imaging tests for the diagnosis of COVID-19.Cochrane Database Syst Rev. 2020 Sep 30;9:CD013639. doi: 10.1002/14651858.CD013639.pub2. Cochrane Database Syst Rev. 2020. Update in: Cochrane Database Syst Rev. 2020 Nov 26;11:CD013639. doi: 10.1002/14651858.CD013639.pub3. PMID: 32997361 Updated.
Cited by
-
Role of Imaging in the Management of Patients with SARS-CoV-2 Lung Involvement Admitted to the Emergency Department: A Systematic Review.Diagnostics (Basel). 2023 May 26;13(11):1856. doi: 10.3390/diagnostics13111856. Diagnostics (Basel). 2023. PMID: 37296708 Free PMC article. Review.
-
A Pictorial Review of the Role of Imaging in the Detection, Management, Histopathological Correlations, and Complications of COVID-19 Pneumonia.Diagnostics (Basel). 2021 Mar 4;11(3):437. doi: 10.3390/diagnostics11030437. Diagnostics (Basel). 2021. PMID: 33806423 Free PMC article. Review.
-
Two episodes of acute dyspnea that were induced by COVID-19 in a peritoneal dialysis patient.CEN Case Rep. 2022 Feb;11(1):22-25. doi: 10.1007/s13730-021-00629-3. Epub 2021 Jul 16. CEN Case Rep. 2022. PMID: 34269997 Free PMC article.
-
Radiation Recall Pneumonitis: The Open Challenge in Differential Diagnosis of Pneumonia Induced by Oncological Treatments.J Clin Med. 2023 Feb 10;12(4):1442. doi: 10.3390/jcm12041442. J Clin Med. 2023. PMID: 36835977 Free PMC article. Review.
-
Qualitative and semi-quantitative ultrasound assessment in delta and Omicron Covid-19 patients: data from high volume reference center.Infect Agent Cancer. 2023 May 27;18(1):34. doi: 10.1186/s13027-023-00515-w. Infect Agent Cancer. 2023. PMID: 37245026 Free PMC article.
References
-
- Reusken C, Broberg EK, Haagmans B, Meijer A, Corman VM, Papa A, Charrel R, Drosten C, Koopmans M, Leitmeyer K, On Behalf Of E-L, Erli N (2020) Laboratory readiness and response for novel coronavirus (2019-nCoV) in expert laboratories in 30 EU/EEA countries, January 2020. Euro Surveill 25(6). 10.2807/1560-7917.ES.2020.25.6.2000082 - PMC - PubMed

