Morphological study of the integument and corporal skeletal muscles of two psammophilous members of Scincidae (Scincus scincus and Eumeces schneideri)
- PMID: 33165963
- PMCID: PMC7839682
- DOI: 10.1002/jmor.21298
Morphological study of the integument and corporal skeletal muscles of two psammophilous members of Scincidae (Scincus scincus and Eumeces schneideri)
Abstract
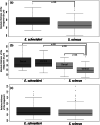
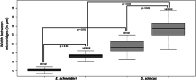
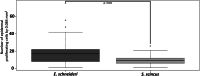
Sand deserts are common biotopes on the earth's surface. Numerous morphological and physiological adaptations have appeared to cope with the peculiar conditions imposed by sandy substrates, such as abrasion, mechanical resistance and the potential low oxygen levels. The psammophilous scincids (Lepidosauria) Scincus scincus and Eumeces schneideri are among those. S. scincus is a species frequently used to study displacement inside a sandy substrate. E. schneideri is a species phylogenetically closely related to S. scincus with a similar lifestyle. The aims of this study focus on the morphology of the integument and the muscular system. Briefly, we describe interspecific differences at the superficial architecture of the scales pattern and the thickness of the integument. We highlight a high cellular turnover rate at the level of the basal germinal layer of the epidermis, which, we suggest, corresponds to an adaptation to cutaneous wear caused by abrasion. We demonstrate the presence of numerous cutaneous holocrine glands whose secretion probably plays a role in the flow of sand along the integument. Several strata of osteoderms strengthen the skin. We characterize the corporal (M. longissimus dorsi and M. rectus abdominus) and caudal muscular fibers using immunohistochemistry, and quantify them using morphometry. The musculature exhibits a high proportion of glycolytic fast fibers that allow rapid burying and are well adapted to this mechanically resistant and oxygen-poor substrate. Oxidative slow fibers are low in abundance, less than 10% in S. scincus, but a little higher in E. schneideri.
Keywords: immunohistochemistry; integument; muscle fibers; sand dwelling; skinks.
© 2020 The Authors. Journal of Morphology published by Wiley Periodicals LLC.
Conflict of interest statement
Both authors declare no conflicts of interest.
Figures





References
-
- Alibardi, L. (2015). Regenerating tail muscles in lizard contain fast but not slow myosin indicating that most myofibers belong to the fast twitch type for rapid contraction. Tissue and Cell, 47(5), 533–540. - PubMed
-
- Alibardi, L. , Maurizii, M. G. , & Taddei, C. (2000). Immunocytochemical and electrophoretic distribution of Cytokeratins in the regenerating epidermis of the lizard Podarcis muralis . Journal of Morphology, 246, 179–191. - PubMed
-
- Alibardi, L. , Maurizii, M. G. , & Taddei, C. (2001). Immunocytochemical and electrophoretic distribution of Cytokeratins in the resting stage epidermis of the lizard Podarcis sicula . Journal of Experimental Zoology, 289, 409–418. - PubMed
-
- Alibardi, L. , & Thompson, M. B. (1999). Epidermal differentiation in the developing scales of embryos of the Australian scincid lizard Lampropholis guichenoti . Journal of Morphology, 241, 139–152. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

