Pharmacokinetic and Pharmacodynamic Profiling of Minocycline for Injection following a Single Infusion in Critically Ill Adults in a Phase IV Open-Label Multicenter Study (ACUMIN)
- PMID: 33168615
- PMCID: PMC8092545
- DOI: 10.1128/AAC.01809-20
Pharmacokinetic and Pharmacodynamic Profiling of Minocycline for Injection following a Single Infusion in Critically Ill Adults in a Phase IV Open-Label Multicenter Study (ACUMIN)
Abstract
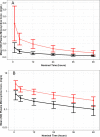
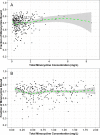
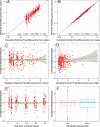
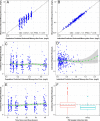
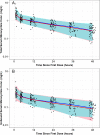
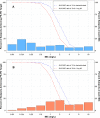
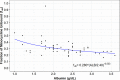
Intravenous (i.v.) minocycline is increasingly used to treat infections caused by multidrug-resistant (MDR) Acinetobacter baumannii Despite its being approved nearly 50 years ago, published information on its pharmacokinetic (PK) profile is limited. This multicenter study examined the PK and probability of pharmacokinetic-pharmacodynamic (PK-PD) target attainment profile of i.v. minocycline in critically ill patients, with suspected or documented infection with Gram-negative bacteria. The PK study population included 55 patients who received a single 200-mg i.v. dose of minocycline. Plasma PK samples were collected predose and 1, 4, 12, 24, 36, and 48 h after initiation of minocycline. Total and unbound minocycline concentrations were determined at each time point. Probabilities of achieving the PK-PD targets associated with stasis and 1-log killing (free area under the curve above the MIC [fAUC:MIC] of 12 and 18, respectively) in an immunocompetent animal pneumonia infection model of A. baumannii were evaluated. A two-compartment population PK model with zero-order i.v. input and first-order elimination, which estimated a constant fraction unbound (fub) for minocycline, best characterized the total and unbound plasma minocycline concentration-time data. The only two covariates retained in the final PK model were body surface area (associated with central volume of distribution) and albumin (associated with fub). In the PK-PD probability of target attainment analyses, minocycline 200 mg i.v. every 12 h (Q12H) was predicted to result in a suboptimal PK-PD profile for patients with A. baumannii infections with MIC values of >1 mg/liter. Like all PK-PD profiling studies of this nature, these findings need clinical confirmation.
Keywords: Acinetobacter; minocycline; pharmacodynamics; pharmacokinetics; population pharmacokinetics.
Copyright © 2021 American Society for Microbiology.
Figures
References
-
- Chmielarczyk A, Pilarczyk-Żurek M, Kamińska W, Pobiega M, Romaniszyn D, Ziółkowski G, Wójkowska-Mach J, Bulanda M. 2016. Molecular epidemiology and drug resistance of Acinetobacter baumannii isolated from hospitals in southern Poland: ICU as a risk factor for XDR strains. Microb Drug Resist 22:328–335. doi:10.1089/mdr.2015.0224. - DOI - PubMed
-
- Centers for Disease Control and Prevention. 2019. Antibiotic resistance threats in the United States 2019. Centers for Disease Control and Prevention, Atlanta, GA. https://www.cdc.gov/drugresistance/pdf/threats-report/2019-ar-threats-re....
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

