Neuropsychological and neuropathological observations of a long-studied case of memory impairment
- PMID: 33168712
- PMCID: PMC7703620
- DOI: 10.1073/pnas.2018960117
Neuropsychological and neuropathological observations of a long-studied case of memory impairment
Abstract
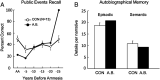
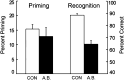
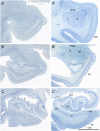
We report neuropsychological and neuropathological findings for a patient (A.B.), who developed memory impairment after a cardiac arrest at age 39. A.B. was a clinical psychologist who, although unable to return to work, was an active participant in our neuropsychological studies for 24 y. He exhibited a moderately severe and circumscribed impairment in the formation of long-term, declarative memory (anterograde amnesia), together with temporally graded retrograde amnesia covering ∼5 y prior to the cardiac arrest. More remote memory for both facts and autobiographical events was intact. His neuropathology was extensive and involved the medial temporal lobe, the diencephalon, cerebral cortex, basal ganglia, and cerebellum. In the hippocampal formation, there was substantial cell loss in the CA1 and CA3 fields, the hilus of the dentate gyrus (with sparing of granule cells), and the entorhinal cortex. There was also cell loss in the CA2 field, but some remnants remained. The amygdala demonstrated substantial neuronal loss, particularly in its deep nuclei. In the thalamus, there was damage and atrophy of the anterior nuclear complex, the mediodorsal nucleus, and the pulvinar. There was also loss of cells in the medial and lateral mammillary nuclei in the hypothalamus. We suggest that the neuropathology resulted from two separate factors: the initial cardiac arrest (and respiratory distress) and the recurrent seizures that followed, which led to additional damage characteristic of temporal lobe epilepsy.
Keywords: amnesia; diencephalon; hippocampus.
Conflict of interest statement
The authors declare no competing interest.
Figures
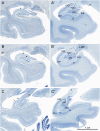


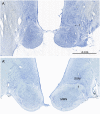
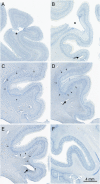
Similar articles
-
Severe amnesia: an usual late complication after temporal lobectomy.Neuropsychologia. 1997 Jul;35(7):975-88. doi: 10.1016/s0028-3932(97)00025-0. Neuropsychologia. 1997. PMID: 9226659
-
The anatomy of amnesia: neurohistological analysis of three new cases.Learn Mem. 2006 Nov-Dec;13(6):699-710. doi: 10.1101/lm.357406. Epub 2006 Nov 13. Learn Mem. 2006. PMID: 17101872 Free PMC article.
-
Retrograde amnesia for facts and events: findings from four new cases.J Neurosci. 1998 May 15;18(10):3943-54. doi: 10.1523/JNEUROSCI.18-10-03943.1998. J Neurosci. 1998. PMID: 9570821 Free PMC article.
-
The case of K.C.: contributions of a memory-impaired person to memory theory.Neuropsychologia. 2005;43(7):989-1021. doi: 10.1016/j.neuropsychologia.2004.10.007. Epub 2005 Jan 12. Neuropsychologia. 2005. PMID: 15769487 Review.
-
EPS Mid-Career Award 2006. Understanding anterograde amnesia: disconnections and hidden lesions.Q J Exp Psychol (Hove). 2008 Oct;61(10):1441-71. doi: 10.1080/17470210802215335. Epub 2008 Jul 31. Q J Exp Psychol (Hove). 2008. PMID: 18671169 Review.
Cited by
-
Post cardiac arrest physical exercise mitigates cell death in the septal and thalamic nuclei and ameliorates contextual fear conditioning deficits in rats.J Cereb Blood Flow Metab. 2023 Mar;43(3):446-459. doi: 10.1177/0271678X221137539. Epub 2022 Nov 11. J Cereb Blood Flow Metab. 2023. PMID: 36369732 Free PMC article.
-
Hypothalamic volume is associated with age, sex and cognitive function across lifespan: a comparative analysis of two large population-based cohort studies.EBioMedicine. 2025 Jan;111:105513. doi: 10.1016/j.ebiom.2024.105513. Epub 2024 Dec 20. EBioMedicine. 2025. PMID: 39708426 Free PMC article.
-
Neuropeptides in the developing human hippocampus under hypoxic-ischemic conditions.J Anat. 2021 Oct;239(4):856-868. doi: 10.1111/joa.13458. Epub 2021 May 24. J Anat. 2021. PMID: 34028021 Free PMC article.
-
Anterior thalamic nuclei: A critical substrate for non-spatial paired-associate memory in rats.Eur J Neurosci. 2022 Oct;56(7):5014-5032. doi: 10.1111/ejn.15802. Epub 2022 Aug 29. Eur J Neurosci. 2022. PMID: 35985792 Free PMC article.
-
In Vivo Microelectrode Arrays for Detecting Multi-Region Epileptic Activities in the Hippocampus in the Latent Period of Rat Model of Temporal Lobe Epilepsy.Micromachines (Basel). 2021 Jun 3;12(6):659. doi: 10.3390/mi12060659. Micromachines (Basel). 2021. PMID: 34205055 Free PMC article.
References
-
- Winslow F., On Obscure Diseases of the Brain and Disorders of the Mind (John W. Davies, London, ed. 2, 1861).
-
- Ribot T., Les Maladies de la Memoire (Appleton-Century-Crofts, New York, 1881) [English translation: Diseases of Memory].
-
- Whitty C. W. M., Zangwill O. L., Amnesia (Butterworths, London, 1966), p. 217.
-
- Milner B., Disorders of learning and memory after temporal lobe lesions in man. Clin. Neurosurg. 19, 421–466 (1972). - PubMed
-
- Squire L. R., The neuropsychology of human memory. Annu. Rev. Neurosci. 5, 241–273 (1982). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

