Skeletal Phenotypes Due to Abnormalities in Mitochondrial Protein Homeostasis and Import
- PMID: 33171986
- PMCID: PMC7664180
- DOI: 10.3390/ijms21218327
Skeletal Phenotypes Due to Abnormalities in Mitochondrial Protein Homeostasis and Import
Abstract
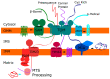
Mitochondrial disease represents a collection of rare genetic disorders caused by mitochondrial dysfunction. These disorders can be quite complex and heterogeneous, and it is recognized that mitochondrial disease can affect any tissue at any age. The reasons for this variability are not well understood. In this review, we develop and expand a subset of mitochondrial diseases including predominantly skeletal phenotypes. Understanding how impairment ofdiverse mitochondrial functions leads to a skeletal phenotype will help diagnose and treat patients with mitochondrial disease and provide additional insight into the growing list of human pathologies associated with mitochondrial dysfunction. The underlying disease genes encode factors involved in various aspects of mitochondrial protein homeostasis, including proteases and chaperones, mitochondrial protein import machinery, mediators of inner mitochondrial membrane lipid homeostasis, and aminoacylation of mitochondrial tRNAs required for translation. We further discuss a complex of frequently associated phenotypes (short stature, cataracts, and cardiomyopathy) potentially explained by alterations to steroidogenesis, a process regulated by mitochondria. Together, these observations provide novel insight into the consequences of impaired mitochondrial protein homeostasis.
Keywords: mitochondrial disease; protein homeostasis; protein import; skeletal abnormality.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Thompson K., Collier J.J., Glasgow R.I.C., Robertson F.M., Pyle A., Blakely E.L., Alston C.L., Oláhová M., McFarland R., Taylor R.W. Recent advances in understanding the molecular genetic basis of mitochondrial disease. J. Inherit. Metab. Dis. 2020;43:36–50. doi: 10.1002/jimd.12104. - DOI - PMC - PubMed
-
- Picard M., Zhang J., Hancock S., Derbeneva O., Golhar R., Golik P., O’Hearn S., Levy S., Potluri P., Lvova M., et al. Progressive increase in mtDNA 3243A>G heteroplasmy causes abrupt transcriptional reprogramming. Proc. Natl. Acad. Sci. USA. 2014;111:E4033–E4042. doi: 10.1073/pnas.1414028111. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

