Studies on the Regulation of (p)ppGpp Metabolism and Its Perturbation Through the Over-Expression of Nudix Hydrolases in Escherichia coli
- PMID: 33178149
- PMCID: PMC7593582
- DOI: 10.3389/fmicb.2020.562804
Studies on the Regulation of (p)ppGpp Metabolism and Its Perturbation Through the Over-Expression of Nudix Hydrolases in Escherichia coli
Abstract
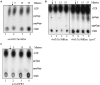
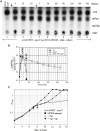
Stringent response mediated by modified guanosine nucleotides is conserved across bacteria and is regulated through the Rel/Spo functions. In Escherichia coli, RelA and SpoT proteins synthesize the modified nucleotides ppGpp and pppGpp, together referred to as (p)ppGpp. SpoT is also the primary (p)ppGpp hydrolase. In this study, using hypomorphic relA alleles, we provide experimental evidence for SpoT-mediated negative regulation of the amplification of RelA-dependent stringent response. We investigated the kinetics of ppGpp degradation in cells recovering from stringent response in the complete absence of SpoT function. We found that, although greatly diminished, there was slow ppGpp degradation and growth resumption after a lag period, concomitant with decrease in ppGpp pool. We present evidence for reduction in the ppGpp degradation rate following an increase in pppGpp pool, during recovery from stringent response. From a genetic screen, the nudix hydrolases MutT and NudG were identified as over-expression suppressors of the growth defect of ΔspoT and ΔspoT ΔgppA strains. The effect of over-expression of these hydrolases on the stringent response to amino acid starvation and basal (p)ppGpp pool was studied. Over-expression of each hydrolase reduced the strength of the stringent response to amino acid starvation, and additionally, perturbed the ratio of ppGpp to pppGpp in strains with reduced SpoT hydrolase activity. In these strains that do not accumulate pppGpp during amino acid starvation, the expression of NudG or MutT supported pppGpp accumulation. This lends support to the idea that a reduction in the SpoT hydrolase activity is sufficient to cause the loss of pppGpp accumulation and therefore the phenomenon is independent of hydrolases that target pppGpp, such as GppA.
Keywords: (p)ppGpp; RelA; SpoT; nudix hydrolases; stringent response.
Copyright © 2020 Sanyal, Vimala and Harinarayanan.
Figures





References
LinkOut - more resources
Full Text Sources

