SWL-1 Reverses Fluconazole Resistance in Candida albicans by Regulating the Glycolytic Pathway
- PMID: 33178156
- PMCID: PMC7596347
- DOI: 10.3389/fmicb.2020.572608
SWL-1 Reverses Fluconazole Resistance in Candida albicans by Regulating the Glycolytic Pathway
Abstract
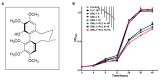
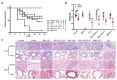
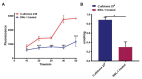
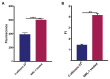
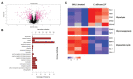
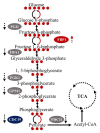
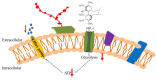
Candida albicans is a ubiquitous clinical fungal pathogen. Prolonged use of the first-line antifungal agent fluconazole (FLC) has intensified fungal resistance and limited its effectiveness for the treatment of fungal infections. The combined administration of drugs has been extensively studied and applied. SWL-1 is a lignin compound derived from the Traditional Chinese Medicine Schisandra chinensis. In this study, we show that SWL-1 reverses resistance to fluconazole in C. albicans when delivered in combination, with a sharp decrease in the IC50 of fluconazole from >200 to 3.74 ± 0.25 μg/ml, and also reverses the fluconazole resistance of C. albicans in vitro, with IC50 from >200 to 5.3 ± 0.3 μg/ml. Moreover, killing kinetics curves confirmed the synergistic effects of fluconazole and SWL-1. Intriguingly, when SWL-1 was administered in combination with fluconazole in a mouse model of systemic infection, the mortality of mice was markedly decreased and fungal colonization of the kidney and lung was reduced. Further mechanistic studies showed that SWL-1 significantly decreased intracellular adenosine 5'-triphosphate (ATP) levels and inhibited the function of the efflux pump responsible for fluconazole resistance of C. albicans. Proteomic analysis of the effects of SWL-1 on C. albicans showed that several enzymes were downregulated in the glycolytic pathway. We speculate that SWL-1 significantly decreased intracellular ATP levels by hindering the glycolysis, and the function of the efflux pump responsible for fluconazole resistance of C. albicans was inhibited, resulting in restoration of fluconazole sensitivity in FLC-resistant C. albicans. This study clarified the effects and mechanism of SWL-1 on C. albicans in vitro and in vivo, providing a novel approach to overcoming fungal resistance.
Keywords: Candida albicans; SWL-1; combination; glycolysis; natural compounds; resistant.
Copyright © 2020 Li, Zhang, Wang, Zhang, Jin, Li, Wang and Xiao.
Figures

References
-
- Alexander B. D., Procop G. W., Dufresne P., Fuller J., Ghannoum M. A., Hanson K. E., et al. (2017). “Clinical and Laboratory Standards Institute (CLSI). Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts. 4th edn. CLSI standard M27”. (Wayne, PA, United States: Clinical and Laboratory Standards Institute; ).
LinkOut - more resources
Full Text Sources

