Asymmetric Mannich reactions of (S)- N- tert-butylsulfinyl-3,3,3-trifluoroacetaldimines with yne nucleophiles
- PMID: 33178357
- PMCID: PMC7607431
- DOI: 10.3762/bjoc.16.217
Asymmetric Mannich reactions of (S)- N- tert-butylsulfinyl-3,3,3-trifluoroacetaldimines with yne nucleophiles
Abstract
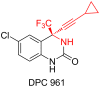
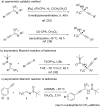
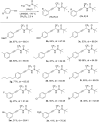
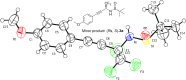
In the present work, arylethynes were studied as new C-nucleophiles in the asymmetric Mannich addition reactions with (S)-N-tert-butylsulfinyl-3,3,3-trifluoroacetaldimine. The reactions were conducted under operationally convenient conditions affording the corresponding Mannich adducts with up to 87% yield and 70:30 diastereoselectivity. The isomeric products can be separated using regular column chromatography to afford diastereomerically pure compounds. The purified Mannich addition products were deprotected to give the target enantiomerically pure trifluoromethylpropargylamines. A mechanistic rationale for the observed stereochemical outcome is discussed.
Keywords: C-nucleophile; CF3-aldimine; arylethynes; asymmetric Mannich reaction; fluorinated propargylamine.
Copyright © 2020, Li et al.; licensee Beilstein-Institut.
Figures
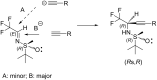
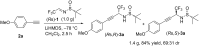
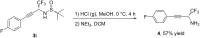
Similar articles
-
Asymmetric synthesis of (3S,1'S)-3-(1-amino-2,2,2-trifluoroethyl)-1-(alkyl)-indolin-2-one derivatives by addition of (S)-N-t-butylsulfinyl-3,3,3-trifluoroacetaldimine to 1-(alkyl)-indolin-2-ones.Org Biomol Chem. 2014 Oct 28;12(40):7909-13. doi: 10.1039/c4ob01453g. Org Biomol Chem. 2014. PMID: 25220306
-
Asymmetric Friedel-Crafts reactions of N-tert-butylsulfinyl-3,3,3-trifluoroacetaldimines: general access to enantiomerically pure indoles containing a 1-amino-2,2,2-trifluoroethyl group.J Org Chem. 2014 Aug 15;79(16):7677-81. doi: 10.1021/jo5012009. Epub 2014 Jul 28. J Org Chem. 2014. PMID: 25051149
-
Asymmetric Mannich reaction between (S)-N-(tert-butanesulfinyl)-3,3,3-trifluoroacetaldimine and malonic acid derivatives. Stereodivergent synthesis of (R)- and (S)-3-amino-4,4,4-trifluorobutanoic acids.Org Biomol Chem. 2014 Mar 7;12(9):1454-62. doi: 10.1039/c3ob42425a. Org Biomol Chem. 2014. PMID: 24445698
-
Synthesis of fluorinated chiral amines using N-tert-butylsulfinyl imines.Future Med Chem. 2009 Aug;1(5):875-88. doi: 10.4155/fmc.09.62. Future Med Chem. 2009. PMID: 21426086 Review.
-
Asymmetric synthesis of α-amino acids via homologation of Ni(II) complexes of glycine Schiff bases. Part 2: aldol, Mannich addition reactions, deracemization and (S) to (R) interconversion of α-amino acids.Amino Acids. 2013 Nov;45(5):1017-33. doi: 10.1007/s00726-013-1580-3. Epub 2013 Sep 17. Amino Acids. 2013. PMID: 24043459 Review.
Cited by
-
Synthesis of Aminoalkyl Sclareolide Derivatives and Antifungal Activity Studies.Molecules. 2023 May 12;28(10):4067. doi: 10.3390/molecules28104067. Molecules. 2023. PMID: 37241807 Free PMC article.
References
LinkOut - more resources
Full Text Sources
