High-dose naloxone: Effects by late administration on pain and hyperalgesia following a human heat injury model. A randomized, double-blind, placebo-controlled, crossover trial with an enriched enrollment design
- PMID: 33180816
- PMCID: PMC7660513
- DOI: 10.1371/journal.pone.0242169
High-dose naloxone: Effects by late administration on pain and hyperalgesia following a human heat injury model. A randomized, double-blind, placebo-controlled, crossover trial with an enriched enrollment design
Abstract
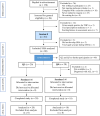
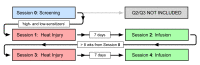
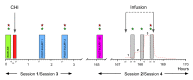
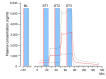
Severe chronic postsurgical pain has a prevalence of 4-10% in the surgical population. The underlying nociceptive mechanisms have not been well characterized. Following the late resolution phase of an inflammatory injury, high-dose μ-opioid-receptor inverse agonists reinstate hypersensitivity to nociceptive stimuli. This unmasking of latent pain sensitization has been a consistent finding in rodents while only observed in a limited number of human volunteers. Latent sensitization could be a potential triggering venue in chronic postsurgical pain. The objective of the present trial was in detail to examine the association between injury-induced secondary hyperalgesia and naloxone-induced unmasking of latent sensitization. Healthy volunteers (n = 80) received a cutaneous heat injury (47°C, 420 s, 12.5 cm2). Baseline secondary hyperalgesia areas were assessed 1 h post-injury. Utilizing an enriched enrollment design, subjects with a magnitude of secondary hyperalgesia areas in the upper quartile ('high-sensitizers' [n = 20]) and the lower quartile ('low-sensitizers' [n = 20]) were selected for further study. In four consecutive experimental sessions (Sessions 1 to 4), the subjects at two sessions (Sessions 1 and 3) received a cutaneous heat injury followed 168 h later (Sessions 2 and 4) by a three-step target-controlled intravenous infusion of naloxone (3.25 mg/kg), or normal saline. Assessments of secondary hyperalgesia areas were made immediately before and stepwise during the infusions. Simple univariate statistics revealed no significant differences in secondary hyperalgesia areas between naloxone and placebo treatments (P = 0.215), or between 'high-sensitizers' and 'low-sensitizers' (P = 0.757). In a mixed-effects model, secondary hyperalgesia areas were significantly larger following naloxone as compared to placebo for 'high-sensitizers' (P < 0.001), but not 'low-sensitizers' (P = 0.651). Although we could not unequivocally demonstrate naloxone-induced reinstatement of heat injury-induced hyperalgesia, further studies in clinical postsurgical pain models are warranted.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

