Transdermal Delivery of Kidney-Targeting Nanoparticles Using Dissolvable Microneedles
- PMID: 33184578
- PMCID: PMC7596160
- DOI: 10.1007/s12195-020-00622-3
Transdermal Delivery of Kidney-Targeting Nanoparticles Using Dissolvable Microneedles
Abstract
Introduction: Chronic kidney disease (CKD) affects approximately 13% of the world's population and will lead to dialysis or kidney transplantation. Unfortunately, clinically available drugs for CKD show limited efficacy and toxic extrarenal side effects. Hence, there is a need to develop targeted delivery systems with enhanced kidney specificity that can also be combined with a patient-compliant administration route for such patients that need extended treatment. Towards this goal, kidney-targeted nanoparticles administered through transdermal microneedles (KNP/MN) is explored in this study.
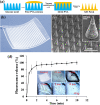
Methods: A KNP/MN patch was developed by incorporating folate-conjugated micelle nanoparticles into polyvinyl alcohol MN patches. Rhodamine B (RhB) was encapsulated into KNP as a model drug and evaluated for biocompatibility and binding with human renal epithelial cells. For MN, skin penetration efficiency was assessed using a Parafilm model, and penetration was imaged via scanning electron microscopy. In vivo, KNP/MN patches were applied on the backs of C57BL/6 wild type mice and biodistribution, organ morphology, and kidney function assessed.
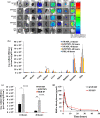
Results: KNP showed high biocompatibility and folate-dependent binding in vitro, validating KNP's targeting to folate receptors in vitro. Upon transdermal administration in vivo, KNP/MN patches dissolved within 30 min. At varying time points up to 48 h post-KNP/MN administration, higher accumulation of KNP was found in kidneys compared with MN that consisted of the non-targeting, control-NP. Histological evaluation demonstrated no signs of tissue damage, and kidney function markers, serum blood urea nitrogen and urine creatinine, were found to be within normal ranges, indicating preservation of kidney health.
Conclusions: Our studies show potential of KNP/MN patches as a non-invasive, self-administrable platform to direct therapies to the kidneys.
Keywords: Chronic kidney disease; Folate; Kidney-targeting; Microneedles; Nanoparticles.
© Biomedical Engineering Society 2020.
Figures





Similar articles
-
Dissolvable microneedles for transdermal drug delivery showing skin pentation and modified drug release.Eur J Pharm Sci. 2023 Mar 1;182:106371. doi: 10.1016/j.ejps.2023.106371. Epub 2023 Jan 5. Eur J Pharm Sci. 2023. PMID: 36621615
-
In vivo studies investigating biodistribution of nanoparticle-encapsulated rhodamine B delivered via dissolving microneedles.J Control Release. 2017 Nov 10;265:57-65. doi: 10.1016/j.jconrel.2017.04.022. Epub 2017 Apr 17. J Control Release. 2017. PMID: 28428065 Free PMC article.
-
Microneedles with Tunable Dissolution Rate.ACS Biomater Sci Eng. 2020 Sep 14;6(9):5061-5068. doi: 10.1021/acsbiomaterials.0c00759. Epub 2020 Aug 19. ACS Biomater Sci Eng. 2020. PMID: 33455299
-
Advances in microneedle-based transdermal delivery for drugs and peptides.Drug Deliv Transl Res. 2022 Jul;12(7):1556-1568. doi: 10.1007/s13346-021-01056-8. Epub 2021 Sep 26. Drug Deliv Transl Res. 2022. PMID: 34564827 Review.
-
Sustainable Drug Release Using Nanoparticle Encapsulated Microneedles.Chem Asian J. 2022 Aug 15;17(16):e202200333. doi: 10.1002/asia.202200333. Epub 2022 Jun 15. Chem Asian J. 2022. PMID: 35644865 Review.
Cited by
-
CD70-Targeted Micelles Enhance HIF2α siRNA Delivery and Inhibit Oncogenic Functions in Patient-Derived Clear Cell Renal Carcinoma Cells.Molecules. 2022 Dec 2;27(23):8457. doi: 10.3390/molecules27238457. Molecules. 2022. PMID: 36500549 Free PMC article.
-
Dissolving Microneedles Developed in Association with Nanosystems: A Scoping Review on the Quality Parameters of These Emerging Systems for Drug or Protein Transdermal Delivery.Pharmaceutics. 2021 Oct 2;13(10):1601. doi: 10.3390/pharmaceutics13101601. Pharmaceutics. 2021. PMID: 34683895 Free PMC article.
-
Oral delivery of nanomedicine for genetic kidney disease.PNAS Nexus. 2024 May 10;3(5):pgae187. doi: 10.1093/pnasnexus/pgae187. eCollection 2024 May. PNAS Nexus. 2024. PMID: 38807632 Free PMC article.
-
Pharmacokinetics and tumor delivery of nanoparticles.J Drug Deliv Sci Technol. 2023 May;83:104404. doi: 10.1016/j.jddst.2023.104404. Epub 2023 Apr 5. J Drug Deliv Sci Technol. 2023. PMID: 38037664 Free PMC article.
-
The Necessity to Investigate In Vivo Fate of Nanoparticle-Loaded Dissolving Microneedles.Pharmaceutics. 2024 Feb 17;16(2):286. doi: 10.3390/pharmaceutics16020286. Pharmaceutics. 2024. PMID: 38399340 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

