Multiple metabolic changes mediate the response of Caenorhabditis elegans to the complex I inhibitor rotenone
- PMID: 33188857
- PMCID: PMC7750303
- DOI: 10.1016/j.tox.2020.152630
Multiple metabolic changes mediate the response of Caenorhabditis elegans to the complex I inhibitor rotenone
Abstract
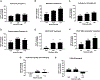
Rotenone, a mitochondrial complex I inhibitor, has been widely used to study the effects of mitochondrial dysfunction on dopaminergic neurons in the context of Parkinson's disease. Although the deleterious effects of rotenone are well documented, we found that young adult Caenorhabditis elegans showed resistance to 24 and 48 h rotenone exposures. To better understand the response to rotenone in C. elegans, we evaluated mitochondrial bioenergetic parameters after 24 and 48 h exposures to 1 μM or 5 μM rotenone. Results suggested upregulation of mitochondrial complexes II and V following rotenone exposure, without major changes in oxygen consumption or steady-state ATP levels after rotenone treatment at the tested concentrations. We found evidence that the glyoxylate pathway (an alternate pathway not present in higher metazoans) was induced by rotenone exposure; gene expression measurements showed increases in mRNA levels for two complex II subunits and for isocitrate lyase, the key glyoxylate pathway enzyme. Targeted metabolomics analyses showed alterations in the levels of organic acids, amino acids, and acylcarnitines, consistent with the metabolic restructuring of cellular bioenergetic pathways including activation of complex II, the glyoxylate pathway, glycolysis, and fatty acid oxidation. This expanded understanding of how C. elegans responds metabolically to complex I inhibition via multiple bioenergetic adaptations, including the glyoxylate pathway, will be useful in interrogating the effects of mitochondrial and bioenergetic stressors and toxicants.
Keywords: Caenorhabditis elegans (C. elegans); Complex I; Glyoxylate; Metabolomics; Mitochondrial metabolism; Rotenone.
Copyright © 2020 Elsevier B.V. All rights reserved.
Conflict of interest statement
Conflict of Interest
The authors declare that they have no conflicts of interest with the contents of this article. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
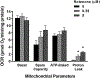
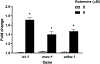
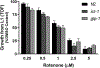

Similar articles
-
Changes in energy metabolism due to acute rotenone-induced mitochondrial complex I dysfunction - An in vivo large animal model.Mitochondrion. 2016 Nov;31:56-62. doi: 10.1016/j.mito.2016.10.003. Epub 2016 Oct 18. Mitochondrion. 2016. PMID: 27769952
-
Partial mitochondrial complex I inhibition induces oxidative damage and perturbs glutamate transport in primary retinal cultures. Relevance to Leber Hereditary Optic Neuropathy (LHON).Neurobiol Dis. 2006 Nov;24(2):308-17. doi: 10.1016/j.nbd.2006.07.016. Epub 2006 Sep 7. Neurobiol Dis. 2006. PMID: 16959493
-
Identification of novel proteins affected by rotenone in mitochondria of dopaminergic cells.BMC Neurosci. 2007 Aug 16;8:67. doi: 10.1186/1471-2202-8-67. BMC Neurosci. 2007. PMID: 17705834 Free PMC article.
-
Evidence for Compartmentalized Axonal Mitochondrial Biogenesis: Mitochondrial DNA Replication Increases in Distal Axons As an Early Response to Parkinson's Disease-Relevant Stress.J Neurosci. 2018 Aug 22;38(34):7505-7515. doi: 10.1523/JNEUROSCI.0541-18.2018. Epub 2018 Jul 20. J Neurosci. 2018. PMID: 30030401 Free PMC article.
-
An adverse outcome pathway for parkinsonian motor deficits associated with mitochondrial complex I inhibition.Arch Toxicol. 2018 Jan;92(1):41-82. doi: 10.1007/s00204-017-2133-4. Epub 2017 Dec 5. Arch Toxicol. 2018. PMID: 29209747 Free PMC article. Review.
Cited by
-
Does treatment with autophagy-enhancers and/or ROS-scavengers alleviate behavioral and neurochemical consequences of low-dose rotenone-induced mild mitochondrial dysfunction in mice?Mol Psychiatry. 2023 Apr;28(4):1667-1678. doi: 10.1038/s41380-023-01955-x. Epub 2023 Jan 23. Mol Psychiatry. 2023. PMID: 36690794 Free PMC article.
-
Chronic high-sugar diet in adulthood protects Caenorhabditis elegans from 6-OHDA-induced dopaminergic neurodegeneration.BMC Biol. 2023 Nov 10;21(1):252. doi: 10.1186/s12915-023-01733-9. BMC Biol. 2023. PMID: 37950228 Free PMC article.
-
Rotenone adaptation promotes migration and invasion of p53-wild-type colon cancer through lipid metabolism.Clin Transl Oncol. 2025 Jun;27(6):2557-2567. doi: 10.1007/s12094-024-03785-x. Epub 2024 Nov 29. Clin Transl Oncol. 2025. PMID: 39612123
-
Regulation of H9C2 cell hypertrophy by 14-3-3η via inhibiting glycolysis.PLoS One. 2024 Jul 22;19(7):e0307696. doi: 10.1371/journal.pone.0307696. eCollection 2024. PLoS One. 2024. PMID: 39038022 Free PMC article.
-
Rescue of a Rotenone Model of Parkinson's Disease in C. elegans by the Mitochondrial Na+/Ca2+ Exchanger Inhibitor CGP37157.Int J Mol Sci. 2025 Apr 4;26(7):3371. doi: 10.3390/ijms26073371. Int J Mol Sci. 2025. PMID: 40244237 Free PMC article.
References
-
- An J, Muoio DM, Shiota M, Fujimoto Y, Cline GW, Shulman GI, Koves TR, Stevens R, Millington D and Newgard CB 2004. Hepatic expression of malonyl-CoA decarboxylase reverses muscle, liver and whole-animal insulin resistance. Nat Med 10, 268–274. - PubMed
-
- Bailey DC, Todt CE, Orfield SE, Denney RD, Snapp IB, Negga R, Montgomery KM, Bailey AC, Pressley AS, Traynor WL and Fitsanakis VA 2016. Caenorhabditis elegans chronically exposed to a Mn/Zn ethylene-bis-dithiocarbamate fungicide show mitochondrial Complex I inhibition and increased reactive oxygen species. NeuroToxicology 56, 170–179. - PubMed
-
- Betarbet R, Sherer TB, MacKenzie G, Garcia-Osuna M, Panov AV and Greenamyre JT 2000. Chronic systemic pesticide exposure reproduces features of Parkinson's disease. Nat Neurosci 3, 1301–1306. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

