Tuning Cancer Fate: Tumor Microenvironment's Role in Cancer Stem Cell Quiescence and Reawakening
- PMID: 33193295
- PMCID: PMC7609361
- DOI: 10.3389/fimmu.2020.02166
Tuning Cancer Fate: Tumor Microenvironment's Role in Cancer Stem Cell Quiescence and Reawakening
Abstract
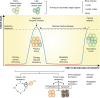
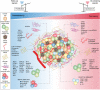
Cancer cell dormancy is a common feature of human tumors and represents a major clinical barrier to the long-term efficacy of anticancer therapies. Dormant cancer cells, either in primary tumors or disseminated in secondary organs, may reawaken and relapse into a more aggressive disease. The mechanisms underpinning dormancy entry and exit strongly resemble those governing cancer cell stemness and include intrinsic and contextual cues. Cellular and molecular components of the tumor microenvironment persistently interact with cancer cells. This dialog is highly dynamic, as it evolves over time and space, strongly cooperates with intrinsic cell nets, and governs cancer cell features (like quiescence and stemness) and fate (survival and outgrowth). Therefore, there is a need for deeper insight into the biology of dormant cancer (stem) cells and the mechanisms regulating the equilibrium quiescence-versus-proliferation are vital in our pursuit of new therapeutic opportunities to prevent cancer from recurring. Here, we review and discuss microenvironmental regulations of cancer dormancy and its parallels with cancer stemness, and offer insights into the therapeutic strategies adopted to prevent a lethal recurrence, by either eradicating resident dormant cancer (stem) cells or maintaining them in a dormant state.
Keywords: cancer stem cells (CSC); disseminated cancer cells (DCC); dormancy; immune escape; immunoediting of cancer; reawakening; tumor evolution; tumor microenvironment.
Copyright © 2020 Sistigu, Musella, Galassi, Vitale and De Maria.
Figures
Similar articles
-
Dormant disseminated tumor cells and cancer stem/progenitor-like cells: Similarities and opportunities.Semin Cancer Biol. 2020 Feb;60:157-165. doi: 10.1016/j.semcancer.2019.09.002. Epub 2019 Sep 3. Semin Cancer Biol. 2020. PMID: 31491559 Review.
-
Tumor dormancy and cancer stem cells: two sides of the same coin?Adv Exp Med Biol. 2013;734:145-79. doi: 10.1007/978-1-4614-1445-2_8. Adv Exp Med Biol. 2013. PMID: 23143979
-
Beyond Tumor Suppression: Senescence in Cancer Stemness and Tumor Dormancy.Cells. 2020 Feb 3;9(2):346. doi: 10.3390/cells9020346. Cells. 2020. PMID: 32028565 Free PMC article. Review.
-
Microenvironmental Regulation of Dormancy in Breast Cancer Metastasis: "An Ally that Changes Allegiances".Adv Exp Med Biol. 2025;1464:373-395. doi: 10.1007/978-3-031-70875-6_18. Adv Exp Med Biol. 2025. PMID: 39821034 Review.
-
Dormancy and cancer stem cells: An enigma for cancer therapeutic targeting.Adv Cancer Res. 2019;141:43-84. doi: 10.1016/bs.acr.2018.12.002. Epub 2019 Jan 16. Adv Cancer Res. 2019. PMID: 30691685 Review.
Cited by
-
Identification and Validation of a Prognostic Signature Derived from the Cancer Stem Cells for Oral Squamous Cell Carcinoma.Int J Mol Sci. 2024 Jan 14;25(2):1031. doi: 10.3390/ijms25021031. Int J Mol Sci. 2024. PMID: 38256104 Free PMC article.
-
Regulation of adult stem cell quiescence and its functions in the maintenance of tissue integrity.Nat Rev Mol Cell Biol. 2023 May;24(5):334-354. doi: 10.1038/s41580-022-00568-6. Epub 2023 Mar 15. Nat Rev Mol Cell Biol. 2023. PMID: 36922629 Free PMC article. Review.
-
Targeting Cancer Stem Cells through Epigenetic Modulation of Interferon Response.J Pers Med. 2022 Apr 1;12(4):556. doi: 10.3390/jpm12040556. J Pers Med. 2022. PMID: 35455671 Free PMC article. Review.
-
Targeting FABP4/UCP2 axis to overcome cetuximab resistance in obesity-driven CRC with drug-tolerant persister cells.Transl Oncol. 2025 Mar;53:102274. doi: 10.1016/j.tranon.2025.102274. Epub 2025 Jan 16. Transl Oncol. 2025. PMID: 39823981 Free PMC article.
-
Epigenomic regulation of stemness contributes to the low immunogenicity of the most mutated human cancer.Cell Rep. 2025 May 27;44(5):115561. doi: 10.1016/j.celrep.2025.115561. Epub 2025 Apr 17. Cell Rep. 2025. PMID: 40250424 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

