Fructose Consumption Affects Glucocorticoid Signaling in the Liver of Young Female Rats
- PMID: 33198224
- PMCID: PMC7698302
- DOI: 10.3390/nu12113470
Fructose Consumption Affects Glucocorticoid Signaling in the Liver of Young Female Rats
Abstract
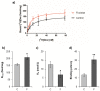
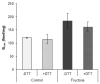
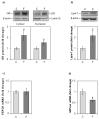
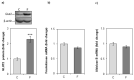
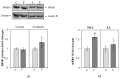
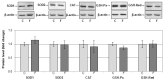
The effects of early-life fructose consumption on hepatic signaling pathways and their relation to the development of metabolic disorders in later life are not fully understood. To investigate whether fructose overconsumption at a young age induces alterations in glucocorticoid signaling that might contribute to development of metabolic disturbances, we analysed glucocorticoid receptor hormone-binding parameters and expression of its target genes involved in gluconeogenesis (phosphoenolpyruvate carboxykinase and glucose-6-phosphatase) and lipid metabolism (lipin-1), as well as redox and inflammatory status in the liver of female rats subjected to a fructose-rich diet immediately after weaning. The fructose diet increased hepatic corticosterone concentration, 11β-hydroxysteroid dehydrogenase type 1 level, glucocorticoid receptor protein level and hormone-binding activity, as well as lipin-1 level. The expression of glucose-6-phosphatase was reduced in fructose-fed rats, while phosphoenolpyruvate carboxykinase remained unaltered. The fructose-rich diet increased the level of fructose transporter GLUT2, while the expression of fructolytic enzymes fructokinase and aldolase B remained unaltered. The diet also affected pro-inflammatory pathways, but had no effect on the antioxidant defence system. In conclusion, a fructose-rich diet applied immediately after weaning promoted lipogenesis and enhanced hepatic glucocorticoid signaling, possibly to protect against inflammatory damage, but without an effect on gluconeogenesis and antioxidant enzymes. Yet, prolonged treatment might ultimately lead to more pronounced metabolic disturbances.
Keywords: antioxidant enzymes; fructose-fed rat; glucocorticoid receptor; inflammation; lipin-1; lipogenesis.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures

Similar articles
-
Enhanced prereceptor glucocorticoid metabolism and lipogenesis impair insulin signaling in the liver of fructose-fed rats.J Nutr Biochem. 2013 Nov;24(11):1790-7. doi: 10.1016/j.jnutbio.2013.04.001. Epub 2013 Jun 14. J Nutr Biochem. 2013. PMID: 23773625
-
Hepatic inflammation induced by high-fructose diet is associated with altered 11βHSD1 expression in the liver of Wistar rats.Eur J Nutr. 2014 Sep;53(6):1393-402. doi: 10.1007/s00394-013-0641-4. Epub 2014 Jan 4. Eur J Nutr. 2014. PMID: 24389792
-
Fructose and stress induce opposite effects on lipid metabolism in the visceral adipose tissue of adult female rats through glucocorticoid action.Eur J Nutr. 2017 Sep;56(6):2115-2128. doi: 10.1007/s00394-016-1251-8. Epub 2016 Jun 21. Eur J Nutr. 2017. PMID: 27324140
-
Fructose-induced inflammation and increased cortisol: A new mechanism for how sugar induces visceral adiposity.Prog Cardiovasc Dis. 2018 May-Jun;61(1):3-9. doi: 10.1016/j.pcad.2017.12.001. Epub 2017 Dec 8. Prog Cardiovasc Dis. 2018. PMID: 29225114 Review.
-
The cross talk of adrenal and Leydig cell steroids in Leydig cells.J Steroid Biochem Mol Biol. 2019 Sep;192:105386. doi: 10.1016/j.jsbmb.2019.105386. Epub 2019 May 29. J Steroid Biochem Mol Biol. 2019. PMID: 31152782 Review.
Cited by
-
Effect of a Fructose-Rich Diet on Gut Microbiota and Immunomodulation: Potential Factors for Multiple Sclerosis.Immunohorizons. 2023 Mar 1;7(3):213-227. doi: 10.4049/immunohorizons.2300008. Immunohorizons. 2023. PMID: 36939622 Free PMC article.
-
Fructose Induces Visceral Adipose Tissue Inflammation and Insulin Resistance Even Without Development of Obesity in Adult Female but Not in Male Rats.Front Nutr. 2021 Nov 11;8:749328. doi: 10.3389/fnut.2021.749328. eCollection 2021. Front Nutr. 2021. PMID: 34869524 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

