CMV-encoded GPCR pUL33 activates CREB and facilitates its recruitment to the MIE locus for efficient viral reactivation
- PMID: 33199520
- PMCID: PMC7860128
- DOI: 10.1242/jcs.254268
CMV-encoded GPCR pUL33 activates CREB and facilitates its recruitment to the MIE locus for efficient viral reactivation
Abstract
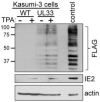
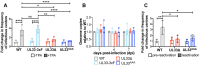
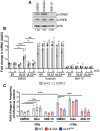
Human cytomegalovirus (HCMV) establishes life-long latent infection in hematopoietic progenitor cells and circulating monocytes in infected individuals. Myeloid differentiation coupled with immune dysregulation leads to viral reactivation, which can cause severe disease and mortality. Reactivation of latent virus requires chromatin reorganization and the removal of transcriptional repressors in exchange for transcriptional activators. While some factors involved in these processes are identified, a complete characterization of the viral and cellular factors involved in their upstream regulation remains elusive. Herein, we show the HCMV-encoded G protein-coupled receptor (GPCR), UL33, is expressed during latency. Although this viral GPCR is not required to maintain latent infection, our data reveal UL33-mediated signaling is important for efficient viral reactivation. Additionally, UL33 signaling induces cellular cyclic AMP response element binding protein (CREB1, referred to here as CREB) phosphorylation, a transcription factor that promotes reactivation when recruited to the major immediate early (MIE) enhancer/promoter. Finally, targeted pharmacological inhibition of CREB activity reverses the reactivation phenotype of the UL33 signaling-deficient mutant. In sum, our data reveal UL33-mediated signaling functions to activate CREB, resulting in successful viral reactivation.
Keywords: CMV; GPCR; Latency reactivation; UL33.
© 2021. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interestsThe authors declare no competing or financial interests.
Figures




Similar articles
-
Host-encoded CTCF regulates human cytomegalovirus latency via chromatin looping.Proc Natl Acad Sci U S A. 2024 Mar 5;121(10):e2315860121. doi: 10.1073/pnas.2315860121. Epub 2024 Feb 26. Proc Natl Acad Sci U S A. 2024. PMID: 38408244 Free PMC article.
-
Activator protein-1 transactivation of the major immediate early locus is a determinant of cytomegalovirus reactivation from latency.Proc Natl Acad Sci U S A. 2020 Aug 25;117(34):20860-20867. doi: 10.1073/pnas.2009420117. Epub 2020 Aug 11. Proc Natl Acad Sci U S A. 2020. PMID: 32788362 Free PMC article.
-
Phorbol ester-induced human cytomegalovirus major immediate-early (MIE) enhancer activation through PKC-delta, CREB, and NF-kappaB desilences MIE gene expression in quiescently infected human pluripotent NTera2 cells.J Virol. 2010 Sep;84(17):8495-508. doi: 10.1128/JVI.00416-10. Epub 2010 May 26. J Virol. 2010. PMID: 20504934 Free PMC article.
-
Role of the cytomegalovirus major immediate early enhancer in acute infection and reactivation from latency.Med Microbiol Immunol. 2008 Jun;197(2):223-31. doi: 10.1007/s00430-007-0069-7. Epub 2007 Dec 19. Med Microbiol Immunol. 2008. PMID: 18097687 Review.
-
Control of Immediate Early Gene Expression for Human Cytomegalovirus Reactivation.Front Cell Infect Microbiol. 2020 Sep 17;10:476. doi: 10.3389/fcimb.2020.00476. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 33072616 Free PMC article. Review.
Cited by
-
Host-encoded CTCF regulates human cytomegalovirus latency via chromatin looping.Proc Natl Acad Sci U S A. 2024 Mar 5;121(10):e2315860121. doi: 10.1073/pnas.2315860121. Epub 2024 Feb 26. Proc Natl Acad Sci U S A. 2024. PMID: 38408244 Free PMC article.
-
The Mouse Cytomegalovirus G Protein-Coupled Receptor Homolog, M33, Coordinates Key Features of In Vivo Infection via Distinct Components of Its Signaling Repertoire.J Virol. 2022 Feb 23;96(4):e0186721. doi: 10.1128/JVI.01867-21. Epub 2021 Dec 8. J Virol. 2022. PMID: 34878888 Free PMC article.
-
Insights into the Transcriptome of Human Cytomegalovirus: A Comprehensive Review.Viruses. 2023 Aug 8;15(8):1703. doi: 10.3390/v15081703. Viruses. 2023. PMID: 37632045 Free PMC article. Review.
-
Why Are Cytomegalovirus-Encoded G-Protein-Coupled Receptors Essential for Infection but Only Variably Conserved?Pathogens. 2025 Mar 3;14(3):245. doi: 10.3390/pathogens14030245. Pathogens. 2025. PMID: 40137730 Free PMC article. Review.
-
HCMV Antivirals and Strategies to Target the Latent Reservoir.Viruses. 2021 May 1;13(5):817. doi: 10.3390/v13050817. Viruses. 2021. PMID: 34062863 Free PMC article. Review.
References
-
- Beisser, P. S., Vink, C., Van Dam, J. G., Grauls, G., Vanherle, S. J. V. and Bruggeman, C. A. (1998). The R33 G protein-coupled receptor gene of rat cytomegalovirus plays an essential role in the pathogenesis of viral infection. J. Virol. 72, 2352-2363. 10.1128/JVI.72.3.2352-2363.1998 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

