Smartphone-Delivered Progressive Muscle Relaxation for the Treatment of Migraine in Primary Care: A Randomized Controlled Trial
- PMID: 33200413
- PMCID: PMC8721526
- DOI: 10.1111/head.14010
Smartphone-Delivered Progressive Muscle Relaxation for the Treatment of Migraine in Primary Care: A Randomized Controlled Trial
Erratum in
-
Erratum.Headache. 2021 Jun;61(6):976. doi: 10.1111/head.14064. Epub 2021 Feb 8. Headache. 2021. PMID: 34214186 No abstract available.
Abstract
Objective: Scalable, accessible forms of behavioral therapy for migraine prevention are needed. We assessed the feasibility and acceptability of progressive muscle relaxation (PMR) delivered by a smartphone application (app) in the Primary Care setting.
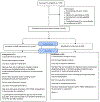
Methods: This pilot study was a non-blinded, randomized, parallel-arm controlled trial of adults with migraine and 4+ headache days/month. Eligible participants spoke English and owned a smartphone. All participants were given the RELAXaHEAD app which includes an electronic headache diary. Participants were randomized to receive 1 of the 2 versions of the app-one with PMR and the other without PMR. The primary outcomes were measures of feasibility (adherence to the intervention and diary entries during the 90-day interval) and acceptability (satisfaction levels). We conducted exploratory analyses to determine whether there was a change in Migraine Disability Assessment Scale (MIDAS) scores or a change in headache days.
Results: Of 139 participants (77 PMR, 62 control), 116 (83%) were female, mean age was 41.7 ± 12.8 years. Most patients 108/139 (78%) had moderate-severe disability. Using a 1-5 Likert scale, participants found the app easy to use (mean 4.2 ± 0.7) and stated that they would be happy to engage in the PMR intervention again (mean 4.3 ± 0.6). For the first 6 weeks, participants practiced PMR 2-4 days/week. Mean per session duration was 11.1 ± 8.3 minutes. Relative to the diary-only group, the PMR group showed a greater non-significant decline in mean MIDAS scores (-8.7 vs -22.7, P = .100) corresponding to a small-moderate mean effect size (Cohen's d = 0.38).
Conclusion: Smartphone-delivered PMR may be an acceptable, accessible form of therapy for migraine. Mean effects show a small-moderate mean effect size in disability scores.
Keywords: behavioral medicine; mHealth; migraine; primary care; relaxation.
© 2020 American Headache Society.
Figures
Similar articles
-
Smartphone based behavioral therapy for pain in multiple sclerosis (MS) patients: A feasibility acceptability randomized controlled study for the treatment of comorbid migraine and ms pain.Mult Scler Relat Disord. 2020 Nov;46:102489. doi: 10.1016/j.msard.2020.102489. Epub 2020 Sep 8. Mult Scler Relat Disord. 2020. PMID: 32950893 Free PMC article. Clinical Trial.
-
User Design and Experience Preferences in a Novel Smartphone Application for Migraine Management: A Think Aloud Study of the RELAXaHEAD Application.Pain Med. 2019 Feb 1;20(2):369-377. doi: 10.1093/pm/pny080. Pain Med. 2019. PMID: 29868895 Free PMC article. Clinical Trial.
-
Introduction of a smartphone based behavioral intervention for migraine in the emergency department.Gen Hosp Psychiatry. 2021 Mar-Apr;69:12-19. doi: 10.1016/j.genhosppsych.2020.12.009. Epub 2021 Jan 8. Gen Hosp Psychiatry. 2021. PMID: 33485090 Free PMC article.
-
Adherence to Behavioral Therapy for Migraine: Knowledge to Date, Mechanisms for Assessing Adherence, and Methods for Improving Adherence.Curr Pain Headache Rep. 2019 Jan 19;23(1):3. doi: 10.1007/s11916-019-0739-3. Curr Pain Headache Rep. 2019. PMID: 30661135 Free PMC article. Review.
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
Cited by
-
Comparing the effect of internet-delivered short-term progressive muscle relaxation and psychoeducation on mindful ability, visceral hypersensitivity and symptoms of patients with irritable bowel syndrome.J Educ Health Promot. 2023 Jul 29;12:259. doi: 10.4103/jehp.jehp_1734_22. eCollection 2023. J Educ Health Promot. 2023. PMID: 37727438 Free PMC article.
-
Incorporating Therapeutic Education and Exercise in Migraine Management: A Biobehavioral Approach.J Clin Med. 2024 Oct 21;13(20):6273. doi: 10.3390/jcm13206273. J Clin Med. 2024. PMID: 39458223 Free PMC article. Review.
-
Digital headache self-management interventions for patients with a primary headache disorder: A systematic review of randomized controlled trials.Headache. 2022 Oct;62(9):1105-1119. doi: 10.1111/head.14392. Headache. 2022. PMID: 36286601 Free PMC article.
-
Behavioral interventions for migraine prevention: A systematic review and meta-analysis.Headache. 2025 Apr;65(4):668-694. doi: 10.1111/head.14914. Epub 2025 Feb 19. Headache. 2025. PMID: 39968795 Free PMC article.
-
The comparative effectiveness of progressive relaxation training on pain characteristics, attack frequency, activity self-efficacy, and pain-related disability in women with episodic tension-type headache and migraine.PLoS One. 2025 Apr 28;20(4):e0320575. doi: 10.1371/journal.pone.0320575. eCollection 2025. PLoS One. 2025. PMID: 40293994 Free PMC article. Clinical Trial.
References
-
- Burch RC, Loder S, Loder E, Smitherman TA. The prevalence and burden of migraine and severe headache in the united states: Updated statistics from government health surveillance studies. Headache. 2015;55:21–34. - PubMed
-
- Campbell J, Penzien D, Wall E. Evidence-Based Guidelines for Migraine Headache: Behavioral and Physical Treatments. St. Paul, MN: American Academy of Neurology; 2000. (US Headache Consortium).
Publication types
MeSH terms
Grants and funding
- UL1 TR001445/TR/NCATS NIH HHS/United States
- R21 AG056920/AG/NIA NIH HHS/United States
- K23 AG049466/AG/NIA NIH HHS/United States
- K23AT009706/AT/NCCIH NIH HHS/United States
- R01 AG048642/AG/NIA NIH HHS/United States
- K23 NS107643/NS/NINDS NIH HHS/United States
- U10 NS077308/NS/NINDS NIH HHS/United States
- R01 AG057531/AG/NIA NIH HHS/United States
- New York University, Center for Healthcare Innovation and Delivery Science (NYU CHIDS)
- NYU Department of Neurology
- P01 AG003949/AG/NIA NIH HHS/United States
- Fellowship to Retain Clinical Scientists/The Doris Duke Charitable Foundation
- R56 AG057548/AG/NIA NIH HHS/United States
- RF1 AG057531/AG/NIA NIH HHS/United States
- RF1 AG054548/AG/NIA NIH HHS/United States
- American Academy of Neurology (AAN) and the American Brain Foundation (ABF)
- R01 AG060933/AG/NIA NIH HHS/United States
- K23 AT009706/AT/NCCIH NIH HHS/United States
- UL1TR001445/New York University Clinical Translational Science Institute (NYU CTSI)
- International Headache Academy (IHA)
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical