A Gain-of-Function Variant in Dopamine D2 Receptor and Progressive Chorea and Dystonia Phenotype
- PMID: 33200438
- PMCID: PMC8049080
- DOI: 10.1002/mds.28385
A Gain-of-Function Variant in Dopamine D2 Receptor and Progressive Chorea and Dystonia Phenotype
Abstract
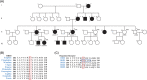
Background: We describe a 4-generation Dutch pedigree with a unique dominantly inherited clinical phenotype of a combined progressive chorea and cervical dystonia carrying a novel heterozygous dopamine D2 receptor (DRD2) variant.
Objectives: The objective of this study was to identify the genetic cause of the disease and to further investigate the functional consequences of the genetic variant.
Methods: After detailed clinical and neurological examination, whole-exome sequencing was performed. Because a novel variant in the DRD2 gene was found as the likely causative gene defect in our pedigree, we sequenced the DRD2 gene in a cohort of 121 Huntington-like cases with unknown genetic cause (Germany). Moreover, functional characterization of the DRD2 variant included arrestin recruitment, G protein activation, and G protein-mediated inhibition of adenylyl cyclase determined in a cell model, and G protein-regulated inward-rectifying potassium channels measured in midbrain slices of mice.
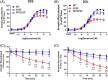
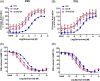
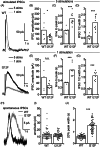
Result: We identified a novel heterozygous variant c.634A > T, p.Ile212Phe in exon 5 of DRD2 that cosegregated with the clinical phenotype. Screening of the German cohort did not reveal additional putative disease-causing variants. We demonstrated that the D2S/L -I212 F receptor exhibited increased agonist potency and constitutive activation of G proteins in human embryonic kidney 239 cells as well as significantly reduced arrestin3 recruitment. We further showed that the D2S -I212 F receptor exhibited aberrant receptor function in mouse midbrain slices.
Conclusions: Our results support an association between the novel p.Ile212Phe variant in DRD2, its modified D2 receptor activity, and the hyperkinetic movement disorder reported in the 4-generation pedigree. © 2020 The Authors. Movement Disorders published by Wiley Periodicals LLC on behalf of International Parkinson and Movement Disorder Society.
Keywords: chorea; dopamine D2 receptor; dystonia; hyperkinetic movement disorder.
© 2020 The Authors. Movement Disorders published by Wiley Periodicals LLC on behalf of International Parkinson and Movement Disorder Society.
Figures
Comment in
-
Reply to: "Childhood Onset Chorea Caused by a Recurrent De Novo DRD2 Variant".Mov Disord. 2021 Jun;36(6):1473-1474. doi: 10.1002/mds.28635. Mov Disord. 2021. PMID: 34145634 No abstract available.
-
Childhood-Onset Chorea Caused by a Recurrent De Novo DRD2 Variant.Mov Disord. 2021 Jun;36(6):1472-1473. doi: 10.1002/mds.28634. Mov Disord. 2021. PMID: 34145635 No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

