Histological, immunohistochemical and transcriptomic characterization of human tracheoesophageal fistulas
- PMID: 33201890
- PMCID: PMC7671559
- DOI: 10.1371/journal.pone.0242167
Histological, immunohistochemical and transcriptomic characterization of human tracheoesophageal fistulas
Abstract
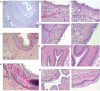
Esophageal atresia (EA) and tracheoesophageal fistula (TEF) are relatively frequently occurring foregut malformations. EA/TEF is thought to have a strong genetic component. Not much is known regarding the biological processes disturbed or which cell type is affected in patients. This hampers the detection of the responsible culprits (genetic or environmental) for the origin of these congenital anatomical malformations. Therefore, we examined gene expression patterns in the TEF and compared them to the patterns in esophageal, tracheal and lung control samples. We studied tissue organization and key proteins using immunohistochemistry. There were clear differences between TEF and control samples. Based on the number of differentially expressed genes as well as histological characteristics, TEFs were most similar to normal esophagus. The BMP-signaling pathway, actin cytoskeleton and extracellular matrix pathways are downregulated in TEF. Genes involved in smooth muscle contraction are overexpressed in TEF compared to esophagus as well as trachea. These enriched pathways indicate myofibroblast activated fibrosis. TEF represents a specific tissue type with large contributions of intestinal smooth muscle cells and neurons. All major cell types present in esophagus are present-albeit often structurally disorganized-in TEF, indicating that its etiology should not be sought in cell fate specification.
Conflict of interest statement
None of the authors have financial, professional, or personal conflicts of interest, all authors reviewed and approved the final manuscript.
Figures


References
-
- del Rosario JF, Orenstein SR. Common pediatric esophageal disorders. Gastroenterologist. 1998;6(2):104–21. - PubMed
-
- Choinitzki V, Zwink N, Bartels E, Baudisch F, Boemers TM, Holscher A, et al. Second study on the recurrence risk of isolated esophageal atresia with or without trachea-esophageal fistula among first-degree relatives: no evidence for increased risk of recurrence of EA/TEF or for malformations of the VATER/VACTERL association spectrum. Birth Defects Res A Clin Mol Teratol. 2013;97(12):786–91. 10.1002/bdra.23205 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

