Pharmacokinetics and Optimal Dose Selection of Cefazolin for Surgical Prophylaxis of Pediatric Patients
- PMID: 33202066
- PMCID: PMC8049034
- DOI: 10.1002/jcph.1785
Pharmacokinetics and Optimal Dose Selection of Cefazolin for Surgical Prophylaxis of Pediatric Patients
Abstract
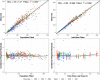
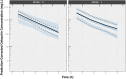
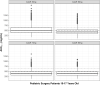
Cefazolin is an antibiotic frequently used for perioperative prophylaxis. Data from healthy adults and pediatric surgery patients were pooled to refine a previously developed population pharmacokinetic (PK) model and to determine the optimal body weight cutoff for selecting fixed doses of either 1 or 2 g cefazolin to produce exposures in pediatric surgery patients similar to a single 2-g dose in adults. Regardless of dose used, cefazolin was well tolerated in pediatric patients. A total of 1102 plasma samples from 62 patients from 3 studies were available to assess the previous model. The pooled data set allowed for simplification of the model such that allometrically scaled clearance and volume parameters were found to provide a robust fit while removing unnecessary covariate relationships. Monte Carlo simulations using the final cefazolin population PK model suggested an optimal weight cutoff of 50 kg, in contrast to the previously suggested 60 kg for a single 2-g dose. Patients at or above this 50-kg cutoff would receive a 2-g dose of cefazolin, and those below 50 kg but ≥25 kg would receive a 1-g dose of cefazolin.
Keywords: cefazolin; model-based simulations; pharmacokinetics; surgical prophylaxis.
© 2020 B. Braun Medical Inc. The Journal of Clinical Pharmacology published by Wiley Periodicals LLC on behalf of American College of Clinical Pharmacology.
Conflict of interest statement
Diana Valencia Martinez, Diane Licursi, Angela Karpf, and Wes Cetnarowski are employees of B. Braun Medical Inc. Michael Schmitz was a principal investigator for Studies HC‐G‐H‐0906 and HC‐G‐H‐1601, and his institution was paid by B. Braun for that participation. At the time this study was conducted, Christopher Rubino and Nikolas Onufrak were employees of Institute for Clinical Pharmacodynamics, Inc. which received consulting fees from B. Braun Medical Inc.
Figures


References
-
- Cefazolin sodium [package insert]. Bethlehem, PA: B. Braun Medical Inc.; 2018.
-
- Bratzler DW, Dellenger EP, Olsen KM, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health‐Syst Pharm. 2013;70(3):195‐283. - PubMed
-
- Woods RK, Dellinger EP. Current guidelines for antibiotic prophylaxis of surgical wounds. Am Fam Physician. 1998;57(11):2731‐2740. - PubMed
-
- Engelman R, Shahian D, Shemin R, et al. The Society of Thoracic Surgeons practice guideline series: antibiotic prophylaxis in cardiac surgery, part II: antibiotic choice. Ann Thorac Surg. 2007;83(4):1569‐1576. - PubMed
-
- Schmitz ML, Blumer JL, Cetnarowski W, Rubino CM. Determination of appropriate weight‐based cutoffs for empiric cefazolin using data from a phase 1 pharmacokinetics and safety study of cefazolin administered for surgical prophylaxis in pediatric patients aged 10 to 12 years. Antimicrob Agents Chemother. 2015;59(7):4173‐4180. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

