Biliary Tract Cancers: Molecular Heterogeneity and New Treatment Options
- PMID: 33202975
- PMCID: PMC7696875
- DOI: 10.3390/cancers12113370
Biliary Tract Cancers: Molecular Heterogeneity and New Treatment Options
Abstract
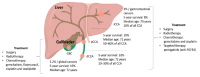
Most patients with biliary tract cancer (BTC) are diagnosed with advanced disease, relapse rates are high in those undergoing surgery and prognosis remains poor, while the incidence is increasing. Treatment options are limited, and chemotherapy is still the standard of care in both adjuvant and advanced disease setting. In recent years, different subtypes of BTC have been defined depending on the anatomical location and genetic and/or epigenetic aberrations. Especially for intrahepatic cholangiocarcinoma (iCCA) novel therapeutic targets have been identified, including fibroblast growth factor receptor 2 gene fusions and isocitrate dehydrogenase 1 and 2 mutations, with molecularly targeted agents having shown evidence of activity in this subgroup of patients. Additionally, other pathways are being evaluated in both iCCA and other subtypes of BTC, alongside targeting of the immune microenvironment. The growing knowledge of BTC biology and molecular heterogeneity has paved the way for the development of new therapeutic approaches that will completely change the treatment paradigm for this disease in the near future. This review provides an overview of the molecular heterogeneity of BTC and summarizes new targets and emerging therapies in development. We also discuss resistance mechanisms, open issues, and future perspectives in the management of BTC.
Keywords: FGFR; IDH; biliary tract cancer; cholangiocarcinoma; molecular characterization; targeted agents; tumor heterogeneity.
Conflict of interest statement
N.P. reports receiving consulting fees from Amgen, Merck Serono, Servier; lectures fees from AbbVie, Gilead, Lilly; travel expenses from Amgen, ArQule; and institutional research funding from Basilea, Merck Serono, Servier. A.L. reports receiving consulting fees from Intercept Pharma, AlfaSigma, AbbVie, Gilead, and MSD. T.P. reports receiving institutional research funding from Lilly. F.C. reports receiving lectures fees from Intercept Pharma. D.J.P. reports receiving consulting fees for Mina Therapeutics, EISAI, Roche, AstraZeneca; lecture fees from ViiV Healthcare, Bayer Healthcare; travel expenses from BMS and Bayer Healthcare; institutional research funding from MSD and BMS. L.R. reports receiving consulting fees from Amgen, ArQule, AstraZeneca, Basilea, Bayer, Celgene, Eisai, Exelixis, Hengrui, Incyte, Ipsen, Lilly, MSD, Nerviano Medical Sciences, Roche, Sanofi; lectures fees from AbbVie, Amgen, Eisai, Gilead, Incyte, Ipsen, Lilly, Roche, Sanofi; travel expenses from Ipsen; and institutional research funding from Agios, ARMO BioSciences, AstraZeneca, BeiGene, Eisai, Exelixis, Fibrogen, Incyte, Ipsen, Lilly, MSD, Nerviano Medical Sciences, Roche. There are no other personal or financial conflicts of interest to disclose.
Figures

References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

