Cetuximab Plus Chemotherapy versus Chemotherapy Alone in Recurrent or Metastatic Head and Neck Squamous Cell Carcinoma: A Cost-Effectiveness Analysis
- PMID: 33204154
- PMCID: PMC7661788
- DOI: 10.2147/CMAR.S272149
Cetuximab Plus Chemotherapy versus Chemotherapy Alone in Recurrent or Metastatic Head and Neck Squamous Cell Carcinoma: A Cost-Effectiveness Analysis
Abstract
Objective: The EXTREME clinical trial revealed that cetuximab plus chemotherapy improved the overall survival time of patients with recurrent or metastatic head and neck squamous cell carcinoma (R/M HNSCC) versus chemotherapy alone. The current study examined the cost-effectiveness of cetuximab plus chemotherapy compared with chemotherapy alone in HNSCC patients from the perspective of China.
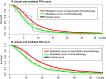
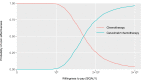
Materials and methods: A partitioned survival model was implemented for R/M HNSCC patients. Survival information was derived from the CHANGE-2 trial. The model was designed as a ten-year time horizon, a 3-week cycle, and a 3% discount rate for costs and utilities. An incremental cost-effectiveness ratio (ICER) value is less than $30,201/quality-adjusted life-year (QALY) was considered cost-effective in China. We analyzed the uncertainty by performing one-way and probabilistic sensitivity analyses.
Results: In the base-case analysis, we found that the ICER of cetuximab plus chemotherapy compared with chemotherapy alone is $172,702/QALY. The results of one-way sensitivity analysis and probabilistic analysis showed that the fluctuations of each variable in its ranges do not cause ICERs to reach acceptable thresholds.
Conclusion: The current observations suggested that treatment with cetuximab plus chemotherapy is not a cost-effective strategy for R/M HNSCC patients in China at a $30,201 willingness to pay threshold.
Keywords: CHANGE-2; HNSCC; cetuximab; cost-effectiveness; recurrent or metastatic head and neck squamous cell carcinoma.
© 2020 Lang and Dong.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures


References
-
- Hu M, Xie P, Lee NY, et al. Hypoxia with 18F-fluoroerythronitroimidazole integrated positron emission tomography and computed tomography (18F-FETNIM PET/CT) in locoregionally advanced head and neck cancer: hypoxia changes during chemoradiotherapy and impact on clinical outcome. Medicine. 2019;98(40):e17067. doi:10.1097/MD.0000000000017067 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

