Plasma proteomic biomarker signature of age predicts health and life span
- PMID: 33210602
- PMCID: PMC7723412
- DOI: 10.7554/eLife.61073
Plasma proteomic biomarker signature of age predicts health and life span
Abstract
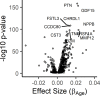
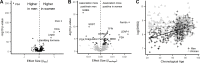
Older age is a strong shared risk factor for many chronic diseases, and there is increasing interest in identifying aging biomarkers. Here, a proteomic analysis of 1301 plasma proteins was conducted in 997 individuals between 21 and 102 years of age. We identified 651 proteins associated with age (506 over-represented, 145 underrepresented with age). Mediation analysis suggested a role for partial cis-epigenetic control of protein expression with age. Of the age-associated proteins, 33.5% and 45.3%, were associated with mortality and multimorbidity, respectively. There was enrichment of proteins associated with inflammation and extracellular matrix as well as senescence-associated secretory proteins. A 76-protein proteomic age signature predicted accumulation of chronic diseases and all-cause mortality. These data support the use of proteomic biomarkers to monitor aging trajectories and to identify individuals at higher risk of disease to be targeted for in depth diagnostic procedures and early interventions.
Keywords: aging; biomarker; computational biology; epidemiology; global health; healthspan; human; proteomics; systems biology.
Conflict of interest statement
TT, NB, GF, JC, AM, AB, BS, SB, LF No competing interests declared
Figures









References
-
- ActiFE study group. Rothenbacher D, Dallmeier D, Christow H, Koenig W, Denkinger M, Klenk J. Association of growth differentiation factor 15 with other key biomarkers, functional parameters and mortality in community-dwelling older adults. Age and Ageing. 2019;48:541–546. doi: 10.1093/ageing/afz022. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

