The potential effects of clinical antidiabetic agents on SARS-CoV-2
- PMID: 33210826
- PMCID: PMC7753367
- DOI: 10.1111/1753-0407.13135
The potential effects of clinical antidiabetic agents on SARS-CoV-2
Abstract
Background: Coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), is currently posing significant threats to public health worldwide. It is notable that a substantial proportion of patients with sever COVID-19 have coexisting diabetic conditions, indicating the progression and outcome of COVID-19 may relate to diabetes. However, it is still unclear whether diabetic treatment principles can be used for the treatment of COVID-19.
Methods: We conducted a computational approach to screen all commonly used clinical oral hypoglycemic drugs to identify the potential inhibitors for the main protease (Mpro ) of SARS-CoV-2, which is one of the key drug targets for anti-COVID-19 drug discovery.
Results: Six antidiabetic drugs with docking scores higher than 8.0 (cutoff value), including repaglinide, canagliflozin, glipizide, gliquidone, glimepiride, and linagliptin, were predicted as the promising inhibitors of Mpro . Interestingly, repaglinide, one of the six antidiabetic drugs with the highest docking score for Mpro , was similar to a previously predicted active molecule nelfinavir, which is a potential anti-HIV and anti-COVID-19 drug. Moreover, we found repaglinide shared similar docking pose and pharmacophores with a reported ligand (N3 inhibitor) and nelfinavir, demonstrating that repaglinide would interact with Mpro in a similar way.
Conclusion: These results indicated that these six antidiabetic drugs may have an extra effect on the treatment of COVID-19, although further studies are necessary to confirm these findings.
背景: 由新型冠状病毒SARS-CoV-2引发的新型冠状病毒肺炎(COVID-19)在全球范围内爆发, 严重危害公众健康。值得注意的是, 相当大比例的COVID-19重症患者同时合并糖尿病, 提示COVID-19的进展和转归可能与糖尿病有关。然而, 目前尚不清楚既往的糖尿病治疗原则能否适用于COVID-19合并糖尿病患者的治疗。 方法: 我们采用计算机模拟方法, 筛选所有临床常用口服降糖药物, 以确定其对COVID-19药物研发关键靶点之一SARS-CoV-2主要蛋白酶(Mpro)的潜在抑制作用。 结果: 在所筛选的口服降糖药物中, 我们发现6种药物的对接分数高于8.0(切点), 包括瑞格列奈, 卡格列净, 格列吡嗪, 格列喹酮, 格列美脲和利格列汀, 提示它们均为潜在的Mpro抑制剂。对接分数最高的瑞格列奈与一种潜在的抗HIV和抗COVID-19药物--奈非那韦得分相近。此外, 我们发现瑞格列奈与已报道的Mpro配体(N3抑制剂)及奈非那韦具有相似的对接姿势和药效基团, 表明瑞格列奈与Mpro间的作用方式可能与N3抑制剂及奈非那韦相似。 结论: 我们的结果提示以上6种降糖药物可能对COVID-19有潜在的治疗作用, 但这些结果尚有待进一步的研究予以证实。.
Keywords: COVID-19; SARS-CoV-2; antidiabetic agents; diabetes; 新型冠状病毒。; 新型冠状病毒肺炎; 糖尿病; 降糖药物.
© 2020 Ruijin Hospital, Shanghai Jiaotong University School of Medicine and John Wiley & Sons Australia, Ltd.
Figures

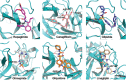
Similar articles
-
In silico analysis of phytochemicals as potential inhibitors of proteases involved in SARS-CoV-2 infection.J Biomol Struct Dyn. 2022 Jul;40(11):5053-5059. doi: 10.1080/07391102.2020.1866669. Epub 2020 Dec 29. J Biomol Struct Dyn. 2022. PMID: 33372574 Free PMC article.
-
In Silico Evaluation of Prospective Anti-COVID-19 Drug Candidates as Potential SARS-CoV-2 Main Protease Inhibitors.Protein J. 2021 Jun;40(3):296-309. doi: 10.1007/s10930-020-09945-6. Epub 2021 Jan 2. Protein J. 2021. PMID: 33387249 Free PMC article.
-
A multi-stage virtual screening of FDA-approved drugs reveals potential inhibitors of SARS-CoV-2 main protease.J Biomol Struct Dyn. 2022 Mar;40(5):2327-2338. doi: 10.1080/07391102.2020.1837680. Epub 2020 Oct 23. J Biomol Struct Dyn. 2022. PMID: 33094680 Free PMC article.
-
Allosteric Binding Sites of the SARS-CoV-2 Main Protease: Potential Targets for Broad-Spectrum Anti-Coronavirus Agents.Drug Des Devel Ther. 2022 Aug 2;16:2463-2478. doi: 10.2147/DDDT.S370574. eCollection 2022. Drug Des Devel Ther. 2022. PMID: 35941927 Free PMC article. Review.
-
An Updated Review on SARS-CoV-2 Main Proteinase (MPro): Protein Structure and Small-Molecule Inhibitors.Curr Top Med Chem. 2021;21(6):442-460. doi: 10.2174/1568026620666201207095117. Curr Top Med Chem. 2021. PMID: 33292134 Review.
Cited by
-
Methodology-Centered Review of Molecular Modeling, Simulation, and Prediction of SARS-CoV-2.Chem Rev. 2022 Jul 13;122(13):11287-11368. doi: 10.1021/acs.chemrev.1c00965. Epub 2022 May 20. Chem Rev. 2022. PMID: 35594413 Free PMC article. Review.
-
Antidiabetic treatment and COVID-19 Outcomes: A population-based cohort study in primary health care in Catalonia during the first wave of the pandemic.Prim Care Diabetes. 2022 Dec;16(6):753-759. doi: 10.1016/j.pcd.2022.10.001. Epub 2022 Oct 4. Prim Care Diabetes. 2022. PMID: 36216752 Free PMC article.
-
The antiviral activity of a small molecule drug targeting the NSP1-ribosome complex against Omicron, especially in elderly patients.Front Cell Infect Microbiol. 2023 Mar 7;13:1141274. doi: 10.3389/fcimb.2023.1141274. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 36960047 Free PMC article.
-
Herbal medications and natural products for patients with covid-19 and diabetes mellitus: Potentials and challenges.Phytomed Plus. 2022 Aug;2(3):100280. doi: 10.1016/j.phyplu.2022.100280. Epub 2022 Apr 18. Phytomed Plus. 2022. PMID: 35463625 Free PMC article. Review.
-
Drug repurposing of argatroban, glimepiride and ranolazine shows anti-SARS-CoV-2 activity via diverse mechanisms.Heliyon. 2025 Jan 10;11(3):e41894. doi: 10.1016/j.heliyon.2025.e41894. eCollection 2025 Feb 15. Heliyon. 2025. PMID: 39968139 Free PMC article.
References
-
- WHO Coronavirus Disease (COVID‐19) Dashboard . 2020. https://covid19.who.int/. Accessed September 7, 2020.
-
- Summary of Probable SARS Cases with Onset of Illness from 1 November 2002 to 31 July 2003. 2003. https://www.who.int/csr/sars/country/table2004_04_21/en/. Accessed February 10, 2020.
MeSH terms
Substances
Grants and funding
- 81925007/The National Science Fund for Distinguished Young Scholars
- 2017YFC1309602/the National Key R&D Program of China
- 2018YFA0507900/the National Key R&D Program of China
- 2016YFC1101100/the National Key R&D Program of China
- 81700714/the National Natural Science Foundation of China
- 81970752/the National Natural Science Foundation of China
- 81471039/the National Natural Science Foundation of China
- 81600673/the National Natural Science Foundation of China
- 2017R013/Talent Project of Third Military Medical University
- 2019XQYYYJ003-2/Talent Project of Third Military Medical University
- 2018YQYLY006/Special Program for Basic Research Frontier of Military Medicine of the Second Affiliated Hospital of Third Military Medical University
- [2016]609/The Distinguished Young Scholars Training Program of the Third Military Medical University
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

