Antitumor effects of novel mAbs against cationic amino acid transporter 1 (CAT1) on human CRC with amplified CAT1 gene
- PMID: 33211385
- PMCID: PMC7894011
- DOI: 10.1111/cas.14741
Antitumor effects of novel mAbs against cationic amino acid transporter 1 (CAT1) on human CRC with amplified CAT1 gene
Abstract
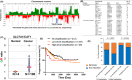
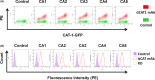
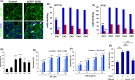
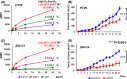
Copy number alterations detected by comparative genomic hybridization (CGH) can lead to the identification of novel cancer-related genes. We analyzed chromosomal aberrations in a set of 100 human primary colorectal cancers (CRCs) using CGH and found a solute carrier (SLC) 7A1 gene, which encodes cationic amino acid transporter 1 (CAT1) with 14 putative transmembrane domains, in a chromosome region (13q12.3) with a high frequency of gene amplifications. SLC7A1/CAT1 is a transporter responsible for the uptake of cationic amino acids (arginine, lysine, and ornithine) essential for cellular growth. Microarray and PCR analyses have revealed that mRNA transcribed from CAT1 is overexpressed in more than 70% of human CRC samples, and RNA interference-mediated knockdown of CAT1 inhibited the cell growth of CRCs. Rats were immunized with rat hepatoma cells expressing CAT1 tagged with green fluorescent protein (GFP), and rat splenocytes were fused with mouse myeloma cells. Five rat monoclonal antibodies (mAbs) (CA1 ~ CA5) reacting with HEK293 cells expressing CAT1-GFP in a GFP expression-dependent manner were selected from established hybridoma clones. Novel anti-CAT1 mAbs selectively reacted with human CRC tumor tissues compared with adjacent normal tissues according to immuno-histochemical staining and bound strongly to numerous human cancer cell lines by flow cytometry. Anti-CAT1 mAbs exhibited internalization activity, antibody-dependent cellular cytotoxicity, and migration inhibition activity against CRC cell lines. Furthermore, CA2 inhibited the in vivo growth of human HT29 and SW-C4 CRC tumors in nude mice. This study suggested CAT1 to be a promising target for mAb therapy against CRCs.
Keywords: CAT1; CRC; SLC7A1; mAb; oncogene addiction.
© 2020 The Authors. Cancer Science published by John Wiley & Sons Australia, Ltd on behalf of Japanese Cancer Association.
Conflict of interest statement
The authors have no conflict of interest to declare.
Figures



References
-
- Fearon ER, Vogelstein B. A genetic model for colorectal tumorigenesis. Cell. 1990;61:759‐767. - PubMed
-
- Sato T, Tanigami A, Yamakawa K, et al. Allelotype of breast cancer cumulative allele losses promote tumor progression in primary breast cancer. Cancer Res. 1990;50:7184‐7189. - PubMed
-
- Barnekow A, Paul E, Schartl M. Expression of the c‐src protooncogene in human skin tumors. Cancer Res. 1987;47:235‐240. - PubMed
-
- Weinstein IB. Disorders in cell circuitry during multistage carcinogenesis: the role of homeostasis. Carcinogenesis. 2000;21:857‐864. - PubMed
-
- Cancer WIB. Addiction to oncogenes–the Achilles heel of cancer. Science. 2002;297:63‐64. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

