Continuous 14 Day Infusional Ifosfamide for Management of Soft-Tissue and Bone Sarcoma: A Single Centre Retrospective Cohort Analysis
- PMID: 33212978
- PMCID: PMC7698576
- DOI: 10.3390/cancers12113408
Continuous 14 Day Infusional Ifosfamide for Management of Soft-Tissue and Bone Sarcoma: A Single Centre Retrospective Cohort Analysis
Abstract
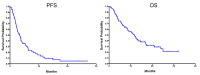
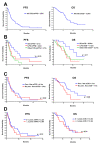
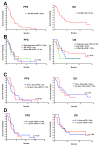
Ifosfamide is used to treat soft-tissue sarcoma (STS) and bone sarcoma (BS), with improved efficacy at doses above 9 g/m2/cycle. To mitigate treatment-associated toxicity with higher doses, continuous infusional ifosfamide is increasingly used. However, clinical outcome data remain limited. Single-centre retrospective analysis of patients treated with four-weekly infusional ifosfamide (14 g/m2/14d) between August 2012 and February 2019 was conducted. Radiological response, progression-free survival (PFS), overall survival (OS) and toxicity were evaluated. Eighty patients were treated-46 with STS and 34 with BS. Patients received a median of three cycles of infusional ifosfamide (1-24). Overall disease control rate (DCR) in STS was 50% (23 of 46 patients), with a median PFS of 3.8 months, and median OS of 13.0 months. In synovial sarcoma (SS), DCR was 80% (12/15), median PFS 8.1 months and median OS 20.9 months. Overall DCR in BS (34 patients) was 30%, with a median PFS of 2.5 months and median OS of 6.2 months. Five patients (6%) stopped treatment due to toxicity alone within the first two cycles. A further 10 patients stopped treatment due to toxicity during later treatment cycles (12%) and 18 patients (23%) required dose modification. Forty-five patients (56%) experienced grade (G) 3/4 haematological toxicity, with 12 episodes of febrile neutropenia and one treatment-related death. Twenty-seven patients (34%) experienced G3/4 non-haematological toxicity, most commonly nausea and vomiting (10, 13%). In summary, infusional ifosfamide has efficacy in STS, most notable in SS. Benefit appears limited in BS. Treatment is associated with toxicity that requires specialist supportive care.
Keywords: bone sarcoma; chemotherapy; infusional ifosfamide; soft-tissue sarcoma.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
High-dose ifosfamide as second- or third-line chemotherapy in refractory bone and soft tissue sarcoma patients.Oncology. 2011;80(3-4):257-61. doi: 10.1159/000328795. Epub 2011 Jul 6. Oncology. 2011. PMID: 21734417
-
High-dose ifosfamide with hematopoietic growth factor support in advanced bone and soft tissue sarcomas.Exp Oncol. 2004 Dec;26(4):320-5. Exp Oncol. 2004. PMID: 15627067 Clinical Trial.
-
Clinical activity and tolerability of a 14-day infusional Ifosfamide schedule in soft-tissue sarcoma.Sarcoma. 2013;2013:868973. doi: 10.1155/2013/868973. Epub 2013 Dec 4. Sarcoma. 2013. PMID: 24369450 Free PMC article.
-
Concurrent ifosfamide-based chemotherapy and irradiation. Analysis of treatment-related toxicity in 43 patients with sarcoma.Cancer. 2001 Sep 15;92(6):1550-5. doi: 10.1002/1097-0142(20010915)92:6<1550::aid-cncr1481>3.0.co;2-c. Cancer. 2001. PMID: 11745234 Review.
-
Current trials and new aspects in soft tissue sarcoma of adults.Cancer Chemother Pharmacol. 2002 May;49 Suppl 1:S4-8. doi: 10.1007/s00280-002-0445-3. Epub 2002 Apr 16. Cancer Chemother Pharmacol. 2002. PMID: 12042982 Review.
Cited by
-
Prolonged 14-day continuous infusion of high-dose ifosfamide for patients with relapsed and refractory high-grade osteosarcoma: a retrospective multicentre cohort study.BMC Cancer. 2024 Jun 19;24(1):747. doi: 10.1186/s12885-024-12498-x. BMC Cancer. 2024. PMID: 38898388 Free PMC article.
-
Chemotherapeutic drugs for soft tissue sarcomas: a review.Front Pharmacol. 2023 Aug 11;14:1199292. doi: 10.3389/fphar.2023.1199292. eCollection 2023. Front Pharmacol. 2023. PMID: 37637411 Free PMC article. Review.
References
-
- Judson I., Verweij J., Gelderblom H., Hartmann J.T., Schöffski P., Blay J.-Y., Kerst J.M., Sufliarsky J., Whelan J., Hohenberger P., et al. Doxorubicin alone versus intensified doxorubicin plus ifosfamide for first-line treatment of advanced or metastatic soft-tissue sarcoma: A randomised controlled phase 3 trial. Lancet Oncol. 2014;15:415–423. doi: 10.1016/S1470-2045(14)70063-4. - DOI - PubMed
LinkOut - more resources
Full Text Sources

