Photodynamic Inactivation of Bacteria with Porphyrin Derivatives: Effect of Charge, Lipophilicity, ROS Generation, and Cellular Uptake on Their Biological Activity In Vitro
- PMID: 33218103
- PMCID: PMC7698881
- DOI: 10.3390/ijms21228716
Photodynamic Inactivation of Bacteria with Porphyrin Derivatives: Effect of Charge, Lipophilicity, ROS Generation, and Cellular Uptake on Their Biological Activity In Vitro
Abstract
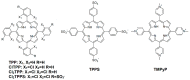
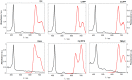
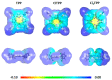
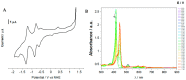
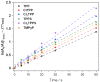
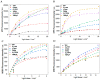
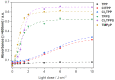
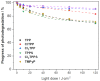
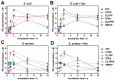
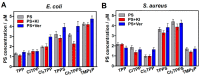
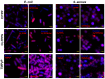
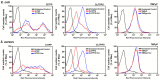
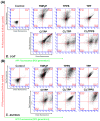
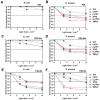
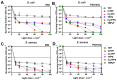
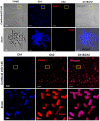
Resistance of microorganisms to antibiotics has led to research on various therapeutic strategies with different mechanisms of action, including photodynamic inactivation (PDI). In this work, we evaluated a cationic, neutral, and anionic meso-tetraphenylporphyrin derivative's ability to inactivate the Gram-negative and Gram-positive bacteria in a planktonic suspension under blue light irradiation. The spectroscopic, physicochemical, redox properties, as well as reactive oxygen species (ROS) generation capacity by a set of photosensitizers varying in lipophilicity were investigated. The theoretical calculations were performed to explain the distribution of the molecular charges in the evaluated compounds. Moreover, logP partition coefficients, cellular uptake, and phototoxicity of the photosensitizers towards bacteria were determined. The role of a specific microbial efflux pump inhibitor, verapamil hydrochloride, in PDI was also studied. The results showed that E. coli exhibited higher resistance to PDI than S. aureus (3-5 logs) with low light doses (1-10 J/cm2). In turn, the prolongation of irradiation (up to 100 J/cm2) remarkably improved the inactivation of pathogens (up to 7 logs) and revealed the importance of photosensitizer photostability. The PDI potentiation occurs after the addition of KI (more than 3 logs extra killing). Verapamil increased the uptake of photosensitizers (especially in E. coli) due to efflux pump inhibition. This effect suggests that PDI is mediated by ROS, the electrostatic charge interaction, and the efflux of photosensitizers (PSs) regulated by multidrug-resistance (MDR) systems. Thus, MDR inhibition combined with PDI gives opportunities to treat more resistant bacteria.
Keywords: antimicrobial activity; efflux pumps; multidrug resistance (MDR); photodynamic inactivation (PDI); porphyrins; reactive oxygen species (ROS); singlet oxygen.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures






Similar articles
-
Properties of halogenated and sulfonated porphyrins relevant for the selection of photosensitizers in anticancer and antimicrobial therapies.PLoS One. 2017 Oct 10;12(10):e0185984. doi: 10.1371/journal.pone.0185984. eCollection 2017. PLoS One. 2017. PMID: 29016698 Free PMC article.
-
Refining antimicrobial photodynamic therapy: effect of charge distribution and central metal ion in fluorinated porphyrins on effective control of planktonic and biofilm bacterial forms.Photochem Photobiol Sci. 2024 Mar;23(3):539-560. doi: 10.1007/s43630-024-00538-1. Epub 2024 Mar 8. Photochem Photobiol Sci. 2024. PMID: 38457119
-
Photodynamic inactivation mediated by 5-aminolevulinic acid of bacteria in planktonic and biofilm forms.Biochem Pharmacol. 2020 Jul;177:114016. doi: 10.1016/j.bcp.2020.114016. Epub 2020 May 7. Biochem Pharmacol. 2020. PMID: 32387459
-
Porphyrinoid photosensitizers mediated photodynamic inactivation against bacteria.Eur J Med Chem. 2019 Aug 1;175:72-106. doi: 10.1016/j.ejmech.2019.04.057. Epub 2019 Apr 29. Eur J Med Chem. 2019. PMID: 31096157 Review.
-
Antimicrobial photodynamic therapy to kill Gram-negative bacteria.Recent Pat Antiinfect Drug Discov. 2013 Aug;8(2):108-20. doi: 10.2174/1574891x113089990012. Recent Pat Antiinfect Drug Discov. 2013. PMID: 23550545 Free PMC article. Review.
Cited by
-
Development of Red-Emissive Porphyrin Graphene Quantum Dots (PGQDs) for Biological Cell-Labeling Applications.ACS Omega. 2022 Oct 19;7(43):38902-38911. doi: 10.1021/acsomega.2c04623. eCollection 2022 Nov 1. ACS Omega. 2022. PMID: 36340159 Free PMC article.
-
Photodynamic Antimicrobial Activity of a Novel 5,10,15,20-Tetrakis (4-Ethylphenyl) Porphyrin against Clinically Important Bacteria.Pharmaceuticals (Basel). 2023 Jul 26;16(8):1059. doi: 10.3390/ph16081059. Pharmaceuticals (Basel). 2023. PMID: 37630978 Free PMC article.
-
meso-Substituted AB3-type phenothiazinyl porphyrins and their indium and zinc complexes photosensitising properties, cytotoxicity and phototoxicity on ovarian cancer cells.RSC Med Chem. 2024 Oct 28;16(2):747-766. doi: 10.1039/d4md00601a. eCollection 2025 Feb 19. RSC Med Chem. 2024. PMID: 39568597 Free PMC article.
-
Red-Shifted Glycoporphyrins: Synthesis via Sonogashira Cross-Coupling and Studies on Reactive Oxygen Species Production.Org Lett. 2025 Jul 25;27(29):7779-7784. doi: 10.1021/acs.orglett.5c02038. Epub 2025 Jul 14. Org Lett. 2025. PMID: 40658819 Free PMC article.
-
Binding mechanism and biological effects of flavone DYRK1A inhibitors for the design of new antidiabetics.Sci Rep. 2023 Oct 23;13(1):18114. doi: 10.1038/s41598-023-44810-3. Sci Rep. 2023. PMID: 37872245 Free PMC article.
References
-
- Kuciauskas D., Lin S., Seely G.R., Moore A.L., Moore A.T.A., Gust D., And T.D., Reed C.A., Boyd P.D.W. Energy and Photoinduced Electron Transfer in Porphyrin−Fullerene Dyads. J. Phys. Chem. 1996;100:15926–15932. doi: 10.1021/jp9612745. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

