Diverse HIV-1 escape pathways from broadly neutralizing antibody PGDM1400 in humanized mice
- PMID: 33218286
- PMCID: PMC7755169
- DOI: 10.1080/19420862.2020.1845908
Diverse HIV-1 escape pathways from broadly neutralizing antibody PGDM1400 in humanized mice
Abstract
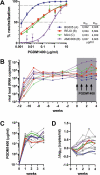
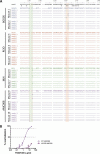
Recent studies have shown the potential of broadly neutralizing antibodies (bnAbs) for HIV-1 treatment. One of the candidate antibodies moving into clinical trials is the bnAb PGDM1400. Here, we studied the therapeutic potency and escape pathways of bnAb PGDM1400 during monovalent therapy in human immune system (HIS) mice using the BG505, REJO, MJ4 and AMC008 virus isolates. PGDM1400 administered during chronic infection caused a modest decrease in viral load in the first week of administration in 7 out of 10 animals, which correlated with the in vitro neutralization sensitivity of the viruses to PGDM1400. As expected for monotherapy, viral loads rebounded after about a week and different viral escape pathways were observed, involving the deletion of glycans in the envelope glycoprotein at positions 130 or 160. (Pre)clinical trials should reveal whether PGDM1400 is a useful component of an antibody combination treatment or as part of a tri-specific antibody.
Keywords: HIV-1; PGDM1400; broadly neutralizing antibody; human immune system mouse; therapeutic; viral escape.
Conflict of interest statement
J.V. was employed by the company AIMM Therapeutics. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. M.J.G and R.W.S are listed on a patent application on PGDM1400.
Figures
References
-
- van der Velden YU, Villaudy J, Siteur-van Rijnstra E, van der Linden CA, Frankin E, Weijer K, Schermer E, Vink MA, Berkhout B, Sanders RW, et al. Short communication: protective efficacy of broadly neutralizing antibody PGDM1400 against HIV-1 challenge in humanized mice. AIDS Res Human Retroviruses. 2018;34(9):790–93. doi:10.1089/aid.2018.0114. - DOI - PubMed
-
- Hessell AJ, Rakasz EG, Poignard P, Hangartner L, Landucci G, Forthal DN, Koff WC, Watkins DI, Burton DR, et al. Broadly neutralizing human anti-HIV antibody 2G12 is effective in protection against mucosal SHIV challenge even at low serum neutralizing titers. PLoS Pathogens. 2009;5(5):e1000433. doi:10.1371/journal.ppat.1000433. - DOI - PMC - PubMed
-
- Deruaz M, Moldt B, Le KM, Power KA, Vrbanac VD, Tanno S, Ghebremichael MS, Allen TM, Tager AM, Burton DR, et al. Protection of humanized mice from repeated intravaginal HIV challenge by passive immunization: a model for studying the efficacy of neutralizing antibodies in vivo. J Infect Dis. 2016;214(4):612–16. doi:10.1093/infdis/jiw203. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
