Myeloma-Modified Adipocytes Exhibit Metabolic Dysfunction and a Senescence-Associated Secretory Phenotype
- PMID: 33218968
- PMCID: PMC7854508
- DOI: 10.1158/0008-5472.CAN-20-1088
Myeloma-Modified Adipocytes Exhibit Metabolic Dysfunction and a Senescence-Associated Secretory Phenotype
Erratum in
-
Correction: Myeloma-Modified Adipocytes Exhibit Metabolic Dysfunction and a Senescence-Associated Secretory Phenotype.Cancer Res. 2025 Nov 14;85(22):4571. doi: 10.1158/0008-5472.CAN-25-3720. Cancer Res. 2025. PMID: 41234128 No abstract available.
Abstract
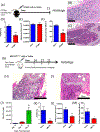
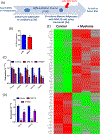
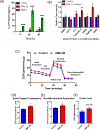
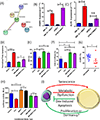
Bone marrow adipocytes (BMAd) have recently been implicated in accelerating bone metastatic cancers, such as acute myelogenous leukemia and breast cancer. Importantly, bone marrow adipose tissue (BMAT) expands with aging and obesity, two key risk factors in multiple myeloma disease prevalence, suggesting that BMAds may influence and be influenced by myeloma cells in the marrow. Here, we provide evidence that reciprocal interactions and cross-regulation of myeloma cells and BMAds play a role in multiple myeloma pathogenesis and treatment response. Bone marrow biopsies from patients with multiple myeloma revealed significant loss of BMAT with myeloma cell infiltration of the marrow, whereas BMAT was restored after treatment for multiple myeloma. Myeloma cells reduced BMAT in different preclinical murine models of multiple myeloma and in vitro using myeloma cell-adipocyte cocultures. In addition, multiple myeloma cells altered adipocyte gene expression and cytokine secretory profiles, which were also associated with bioenergetic changes and induction of a senescent-like phenotype. In vivo, senescence markers were also increased in the bone marrow of tumor-burdened mice. BMAds, in turn, provided resistance to dexamethasone-induced cell-cycle arrest and apoptosis, illuminating a new possible driver of myeloma cell evolution in a drug-resistant clone. Our findings reveal that bidirectional interactions between BMAds and myeloma cells have significant implications for the pathogenesis and treatment of multiple myeloma. Targeting senescence in the BMAd or other bone marrow cells may represent a novel therapeutic approach for treatment of multiple myeloma. SIGNIFICANCE: This study changes the foundational understanding of how cancer cells hijack the bone marrow microenvironment and demonstrates that tumor cells induce senescence and metabolic changes in adipocytes, potentially driving new therapeutic directions.
©2020 American Association for Cancer Research.
Conflict of interest statement
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

