The Interplay Between Beta-Amyloid 1-42 (Aβ1-42)-Induced Hippocampal Inflammatory Response, p-tau, Vascular Pathology, and Their Synergistic Contributions to Neuronal Death and Behavioral Deficits
- PMID: 33224025
- PMCID: PMC7667153
- DOI: 10.3389/fnmol.2020.552073
The Interplay Between Beta-Amyloid 1-42 (Aβ1-42)-Induced Hippocampal Inflammatory Response, p-tau, Vascular Pathology, and Their Synergistic Contributions to Neuronal Death and Behavioral Deficits
Abstract
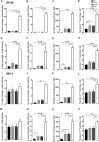
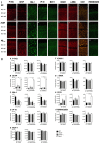
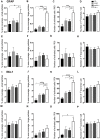
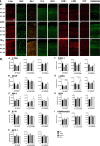
Alzheimer's disease (AD), the most common chronic neurodegenerative disorder, has complex neuropathology. The principal neuropathological hallmarks of the disease are the deposition of extracellular β-amyloid (Aβ) plaques and neurofibrillary tangles (NFTs) comprised of hyperphosphorylated tau (p-tau) protein. These changes occur with neuroinflammation, a compromised blood-brain barrier (BBB) integrity, and neuronal synaptic dysfunction, all of which ultimately lead to neuronal cell loss and cognitive deficits in AD. Aβ1-42 was stereotaxically administered bilaterally into the CA1 region of the hippocampi of 18-month-old male C57BL/6 mice. This study aimed to characterize, utilizing immunohistochemistry and behavioral testing, the spatial and temporal effects of Aβ1-42 on a broad set of parameters characteristic of AD: p-tau, neuroinflammation, vascular pathology, pyramidal cell survival, and behavior. Three days after Aβ1-42 injection and before significant neuronal cell loss was detected, acute neuroinflammatory and vascular responses were observed. These responses included the up-regulation of glial fibrillary acidic protein (GFAP), cell adhesion molecule-1 (PECAM-1, also known as CD31), fibrinogen labeling, and an increased number of activated astrocytes and microglia in the CA1 region of the hippocampus. From day 7, there was significant pyramidal cell loss in the CA1 region of the hippocampus, and by 30 days, significant localized up-regulation of p-tau, GFAP, Iba-1, CD31, and alpha-smooth muscle actin (α-SMA) in the Aβ1-42-injected mice compared with controls. These molecular changes in Aβ1-42-injected mice were accompanied by cognitive deterioration, as demonstrated by long-term spatial memory impairment. This study is reporting a comprehensive examination of a complex set of parameters associated with intrahippocampal administration of Aβ1-42 in mice, their spatiotemporal interactions and combined contribution to the disease progression. We show that a single Aβ injection can reproduce aspects of the inflammatory, vascular, and p-tau induced pathology occurring in the AD human brain that lead to cognitive deficits.
Keywords: Alzheiemer’s disease; cognition; neuroinflamamation; tau phosphorylation; β-amyloid.
Copyright © 2020 Calvo-Flores Guzmán, Chaffey, Palpagama, Waters, Boix, Tate, Peppercorn, Dragunow, Waldvogel, Faull and Kwakowsky.
Figures






References
-
- Baluchnejadmojara D. T., Mohamadi-Zarch S., Roghani M. (2019). Safranal, an active ingredient of saffron, attenuates cognitive deficits in amyloid β-induced rat model of Alzheimer’s disease: underlying mechanisms metabolic brain disease. Metab. Brain Dis. 34, 1747–1759. 10.1073/pnas.0911829107 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

