Knowing When to Self-Eat - Fine-Tuning Autophagy Through ATG8 Iso-forms in Plants
- PMID: 33224169
- PMCID: PMC7669990
- DOI: 10.3389/fpls.2020.579875
Knowing When to Self-Eat - Fine-Tuning Autophagy Through ATG8 Iso-forms in Plants
Abstract
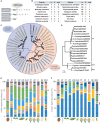
Autophagy is a catabolic process that takes place under both normal and adverse conditions and is important for the degradation of various organelles and proteins that are no longer needed. Thus, it can be viewed as both a constitutive recycling machinery and an adaptation mechanism. Increase in the activity of autophagy can be caused by multiple biotic and abiotic stress factors. Though intensive research in the past decade has elucidated many molecular details of plant autophagy, the mechanisms of induction and regulation of the process remain understudied. Here, we discuss the role of ATG8 proteins in autophagic signaling and regulation with an emphasis on the significance of ATG8 diversification for adapting autophagy to the changing needs of plants.
Keywords: abiotic stress; adaptation mechanism; plant autophagy; recycling; regulation target; regulator.
Copyright © 2020 Boycheva Woltering and Isono.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

