Rat Cytomegalovirus Virion-Associated Proteins R131 and R129 Are Necessary for Infection of Macrophages and Dendritic Cells
- PMID: 33228102
- PMCID: PMC7699341
- DOI: 10.3390/pathogens9110963
Rat Cytomegalovirus Virion-Associated Proteins R131 and R129 Are Necessary for Infection of Macrophages and Dendritic Cells
Abstract
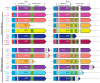
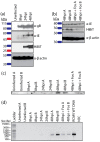
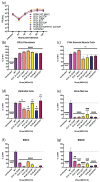
Cytomegalovirus (CMV) establishes persistent, latent infection in hosts, causing diseases in immunocompromised patients, transplant recipients, and neonates. CMV infection modifies the host chemokine axis by modulating chemokine and chemokine receptor expression and by encoding putative chemokine and chemokine receptor homologues. The viral proteins have roles in cellular signaling, migration, and transformation, as well as viral dissemination, tropism, latency and reactivation. Herein, we review the contribution of CMV-encoded chemokines and chemokine receptors to these processes, and further elucidate the viral tropism role of rat CMV (RCMV) R129 and R131. These homologues of the human CMV (HCMV)-encoded chemokines UL128 and UL130 are of particular interest because of their dual role as chemokines and members of the pentameric entry complex, which is required for entry into cell types that are essential for viral transmission and dissemination. The contributions of UL128 and UL130 to acceleration of solid organ transplant chronic rejection are poorly understood, and are in need of an effective in vivo model system to elucidate the phenomenon. We demonstrated similar molecular entry requirements for R129 and R131 in the rat cells, as observed for HCMV, and provided evidence that R129 and R131 are part of the viral entry complex required for entry into macrophages, dendritic cells, and bone marrow cells.
Keywords: chemokine receptors; chemokines; cytomegalovirus; dendritic cells; dissemination; viral entry.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures

References
-
- Johansson I., Andersson R., Friman V., Selimovic N., Hanzen L., Nasic S., Nyström U., Sigurdardottir V. Cytomegalovirus infection and disease reduce 10-year cardiac allograft vasculopathy-free survival in heart transplant recipients. BMC Infect. Dis. 2015;15:582. doi: 10.1186/s12879-015-1321-1. - DOI - PMC - PubMed
-
- Orloff S.L., Hwee Y.-K.K., Kreklywich C., Andoh T.F., Hart E., Smith P.A., Messaoudi I., Streblow D.N. Cytomegalovirus latency promotes cardiac lymphoid neogenesis and accelerated allograft rejection in CMV Naive recipients. Am. J. Transplant. 2011;11:45–55. doi: 10.1111/j.1600-6143.2010.03365.x. - DOI - PMC - PubMed
-
- Russell M.E., Hancock W.W., Wallace A.F., Wyner L.R., Karnovsky M.J. Modulation of inflammatory activation pathways in the Lewis-to-F-344 rat chronic cardiac rejection model. Transplant. Proc. 1995;27:2100–2104. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

