Multiplex Immunoassay Techniques for On-Site Detection of Security Sensitive Toxins
- PMID: 33233770
- PMCID: PMC7699850
- DOI: 10.3390/toxins12110727
Multiplex Immunoassay Techniques for On-Site Detection of Security Sensitive Toxins
Abstract
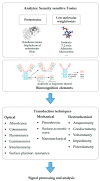
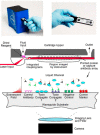
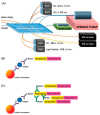
Biological toxins are a heterogeneous group of high molecular as well as low molecular weight toxins produced by living organisms. Due to their physical and logistical properties, biological toxins are very attractive to terrorists for use in acts of bioterrorism. Therefore, among the group of biological toxins, several are categorized as security relevant, e.g., botulinum neurotoxins, staphylococcal enterotoxins, abrin, ricin or saxitoxin. Additionally, several security sensitive toxins also play a major role in natural food poisoning outbreaks. For a prompt response to a potential bioterrorist attack using biological toxins, first responders need reliable, easy-to-use and highly sensitive methodologies for on-site detection of the causative agent. Therefore, the aim of this review is to present on-site immunoassay platforms for multiplex detection of biological toxins. Furthermore, we introduce several commercially available detection technologies specialized for mobile or on-site identification of security sensitive toxins.
Keywords: electrochemical biosensor; low molecular weight toxins; multiplex immunoassay platforms; on-site detection; optical biosensor; proteotoxins; security sensitive toxins.
Conflict of interest statement
C.P. and T.E. are current employees of Bruker Optik GmbH. Bruker Optik GmbH has licensed electrochemical biochip technology according to EP 1200817 B1 from Fraunhofer Institute for Silicon Technology (Itzehoe, Germany) and is manufacturer of the pBDi platform. This does not influence our adherence to all policies on sharing data and materials.
Figures




References
-
- Franz D.R. Textbook of Military Medicine, Part 1: Medical Aspects of Chemical and Biological Warfare. Office of The Surgeon General at TMM Publications, Department of the Army; Washington, DC, USA: 1997. Defense against toxin weapons; pp. 603–619.
-
- The Australia Group-Biological Agents. [(accessed on 21 October 2020)]; Available online: https://www.dfat.gov.au/publications/minisite/theaustraliagroupnet/site/....
-
- Organization for the Prohibition of Chemical Weapons Scientific, Advisory Board SAB-II Annex 1. [(accessed on 19 November 2020)];1999 Available online: https://www.opcw.org/resources/documents/subsidiary-bodies/scientific-ad....
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

