Smartphone measurements of physical activity and fitness are associated with early trial discontinuation of patients in (hemato)oncology phase I/II clinical trials
- PMID: 33236210
- PMCID: PMC8163675
- DOI: 10.1007/s00520-020-05902-2
Smartphone measurements of physical activity and fitness are associated with early trial discontinuation of patients in (hemato)oncology phase I/II clinical trials
Abstract
Background: Patients, who discontinue early, do not benefit from phase I/II clinical trials (early-phase clinical trials (EPCT)). In this study, associations between objective smartphone measurements of physical activity and fitness and early trial discontinuation in patients with cancer participating in EPCT were investigated.
Methods: Before start of treatment, physical activity (steps/day) and physical fitness (meters walked in 6 min) were measured with a smartphone, and patient-reported physical function (PRO-PF) was assessed (EORTC QLQ-C30-PF). Early trial discontinuation was defined as discontinuation ≤ 28 days. Univariable logistic regression analyses were performed to study associations of physical activity, fitness, and function with early trial discontinuation. Optimal cutoff values of physical activity and fitness were assessed with ROCs, based on positive predictive values (PPV).
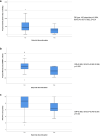
Results: Median (interquartile range (IQR)) step count was 4263 (2548-6897) steps/day, mean ± standard deviation 6-min walking distance was 477 ± 120 m and median (IQR) PRO-PF score was 83 (67-95) points. Fourteen patients (12%) discontinued the trial early. Smartphone measurements of physical activity in units of 100 steps per day (odds ratio (OR) = 0.96, 95% CI = 0.94-0.99, p = 0.01), physical fitness (OR = 0.99, 95% CI = 0.98-0.99, p < 0.01), and PRO-PF (OR = 0.97, 95% CI = 0.94-1.00, p = 0.03) were associated with early trial discontinuation. Optimal cutoff values were < 900 steps for physical activity and < 285 m for physical fitness. PPV for early trial discontinuation was 100% in patients who walked both < 1500 steps per day and < 300 m in 6 min.
Conclusions: Objective smartphone measurements of physical activity and fitness are associated with early trial discontinuation. However, cutoff values should be externally validated in a larger cohort before implementation in clinical practice.
Keywords: Early trial discontinuation; Physical activity; Physical fitness; Smartphone measurements.
Conflict of interest statement
The authors declare that they have no conflict of interest. We have full control of all primary data and we agree to allow the journal to review the data if requested.
Figures
Similar articles
-
Decline in Smartphone-Assessed Physical Activity Level is Associated With Clinical Outcomes in Phase I/II Clinical Cancer Trials.J Natl Compr Canc Netw. 2024 May 22;22(5):323-330. doi: 10.6004/jnccn.2024.7001. J Natl Compr Canc Netw. 2024. PMID: 38776960
-
Feasibility, validity and reliability of objective smartphone measurements of physical activity and fitness in patients with cancer.BMC Cancer. 2018 Oct 29;18(1):1052. doi: 10.1186/s12885-018-4983-4. BMC Cancer. 2018. PMID: 30373549 Free PMC article.
-
The STEPWISE study: study protocol for a smartphone-based exercise solution for people with Parkinson's Disease (randomized controlled trial).BMC Neurol. 2023 Sep 12;23(1):323. doi: 10.1186/s12883-023-03355-8. BMC Neurol. 2023. PMID: 37700241 Free PMC article.
-
The effects of step-count monitoring interventions on physical activity: systematic review and meta-analysis of community-based randomised controlled trials in adults.Int J Behav Nutr Phys Act. 2020 Oct 9;17(1):129. doi: 10.1186/s12966-020-01020-8. Int J Behav Nutr Phys Act. 2020. PMID: 33036635 Free PMC article.
-
Digital technology for delivering and monitoring exercise programs for people with cystic fibrosis.Cochrane Database Syst Rev. 2023 Jun 9;6(6):CD014605. doi: 10.1002/14651858.CD014605.pub2. Cochrane Database Syst Rev. 2023. PMID: 37294546 Free PMC article. Review.
Cited by
-
Feasibility and Acceptability of Collecting Passive Smartphone Data for Potential Use in Digital Phenotyping Among Family Caregivers and Patients With Advanced Cancer.JCO Clin Cancer Inform. 2025 Jan;9:e2400201. doi: 10.1200/CCI-24-00201. Epub 2025 Jan 2. JCO Clin Cancer Inform. 2025. PMID: 39746166
References
-
- Bachelot T, Ray-Coquard I, Catimel G, Ardiet C, Guastalla JP, Dumortier A, Chauvin F, Droz JP, Philip T, Clavel M. Multivariable analysis of prognostic factors for toxicity and survival for patients enrolled in phase I clinical trials. Ann Oncol. 2000;11(2):151–156. doi: 10.1023/A:1008368319526. - DOI - PubMed
-
- Olmos D, A’Hern RP, Marsoni S, Morales R, Gomez-Roca C, Verweij J, Voest EE, Schoffski P, Ang JE, Penel N, Schellens JH, Del Conte G, Brunetto AT, Evans TR, Wilson R, Gallerani E, Plummer R, Tabernero J, Soria JC, Kaye SB. Patient selection for oncology phase I trials: a multi-institutional study of prognostic factors. J Clin Oncol. 2012;30(9):996–1004. doi: 10.1200/JCO.2010.34.5074. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

