Duration of seropositivity following yellow fever vaccination in U.S. military service members
- PMID: 33239225
- PMCID: PMC11349173
- DOI: 10.1016/j.vaccine.2020.11.022
Duration of seropositivity following yellow fever vaccination in U.S. military service members
Abstract
Background: The United States military regularly deploys thousands of service members throughout areas of South America and Africa that are endemic for yellow fever (YF) virus. To determine if booster doses might be needed for service members who are repetitively or continually deployed to YF endemic areas, we evaluated seropositivity among US military personnel receiving a single dose of YF vaccine based on time post-vaccination.
Methods: Serum antibodies were measured using a plaque reduction neutralization test with 50% cutoff in 682 military personnel at 5-39 years post-vaccination. We determined noninferiority of immune response by comparing the proportion seropositive among those vaccinated 10-14 years previously with those vaccinated 5-9 years previously. Noninferiority was supported if the lower-bound of the 2-tailed 95% CI for p10-14years - p5-9years was ≥-0.10. Additionally, the geometric mean antibody titer (GMT) at various timepoints following vaccination were compared to the GMT at 5-9 years.
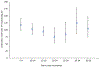
Results: The proportion of military service members with detectable neutralizing antibodies 10-14 years after a single dose of YF vaccine (95.8%, 95% CI 91.2-98.1%) was non-inferior to the proportion 5-9 years after vaccination (97.8%, 95% CI 93.7-99.3%). Additionally, GMT among vaccine recipients at 10-14 years post vaccination (99, 95% CI 82-121) was non-inferior to GMT in YF vaccine recipients at 5-9 years post vaccination (115, 95% CI 96-139). The proportion of vaccinees with neutralizing antibodies remained high, and non-inferior, among those vaccinated 15-19 years prior (98.5%, 95%CI 95.5-99.7%). Although the proportion seropositive decreased among vaccinees ≥ 20 years post vaccination, >90% remained seropositive.
Conclusions: Neutralizing antibodies were present in > 95% of vaccine recipients for at least 19 years after vaccination, suggesting that booster doses every 10 years are not essential for most U.S. military personnel.
Keywords: Military; Seropositivity; Vaccine; Yellow fever.
Published by Elsevier Ltd.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
References
-
- Staples JE, Monath TP, Gersham MD, Barrett ADT. Yellow fever vaccines. In: Plotkin SA, Orenstein WA, Offit PA, Edwards KM, editors. Vaccines. Elsevier; Philadelphia, PA; 2018. p. 1181–267.
-
- Staples JE, Gershman M, Fischer M. Centers for Disease Control and Prevention. Yellow fever vaccine: recommendations of the advisory committee on immunization practices. MMWR Recomm Rep 2010;59:1–27. - PubMed
-
- Lloyd W, Theiler M, Ricci NI. Modification of the virulence of yellow fever virus by cultivation in tissues in vitro. Trans R Soc Trop Med Hyg. 1936;29:481–529.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

